Sonogenetics: Non-Invasive Cellular Engineering
Executive Summary
Sonogenetics is an emerging approach that enables non-invasive modulation of cellular activity using ultrasound in combination with genetically encoded sonosensitive mediators. Established neuromodulation techniques such as deep brain stimulation and optogenetics have advanced neuroscience research but remain limited by invasiveness, restricted tissue penetration and procedural complexity. Ultrasound offers deeper tissue penetration without surgical intervention and has therefore been investigated as an alternative modality for controlling cellular function.
Sonogenetic systems rely on sonosensitive mediators that convert ultrasound-induced thermal or mechanical stimuli into defined cellular responses. These mediators include heat-responsive genetic switches, thermosensitive and mechanosensitive ion channels and engineered proteins capable of responding to membrane deformation or localized temperature changes. Together, these systems enable spatially and temporally controlled modulation of gene expression and neuronal activity in preclinical models.
Current research has explored sonogenetic approaches in areas such as neuromodulation, visual restoration and tumor immunotherapy. Published studies report controlled cellular activation and functional responses under defined conditions. However, most sonogenetic systems remain at an early stage of development, with challenges related to mediator selection, ultrasound parameter optimization, gene delivery and safety.
Overall, the published literature positions sonogenetics as a developing platform for non-invasive cellular engineering, with ongoing research focused on improving reliability, specificity and translational relevance.
1. Introduction
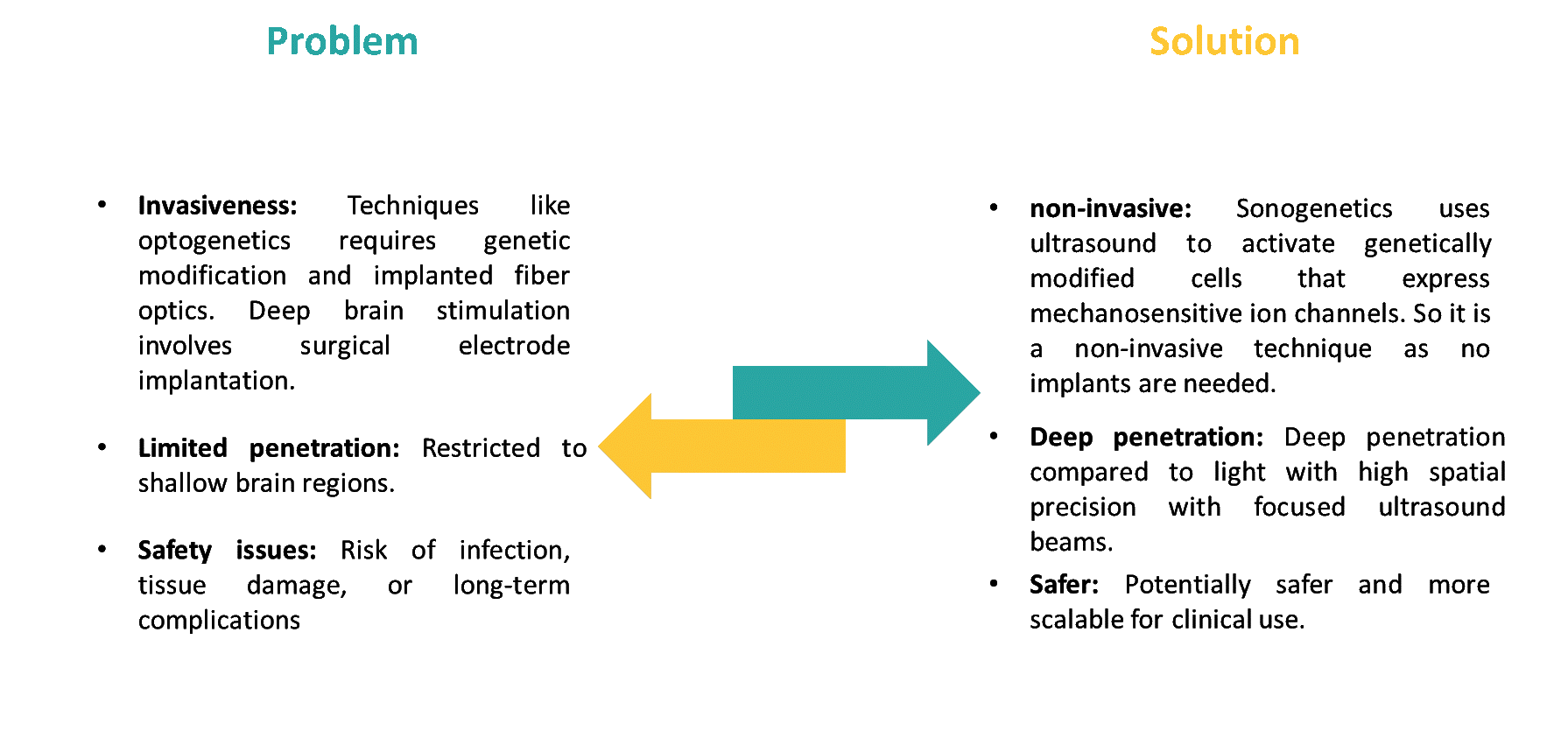
Controlling neurons and other excitable cells with accuracy has reshaped the field of neuroscience and medicine. Well known approaches such as deep brain stimulation and optogenetics have provided remarkable advancements in understanding brain networks and opened new paths for therapy. However, these methods still face challenges such as restricted tissue penetration, invasiveness and complexity. Deep brain stimulation requires surgical implantation of electrodes, carrying risks of infection and long-term complications and optogenetics depends on implanted fiber optics and has shallow penetration of light through biological tissue.
In contrast, sonogenetics has emerged as a promising approach to overcoming these limitations. Sonogenetics refers to the use of ultrasound waves to control cells through sonosensitive mediators (SSMs) that are genetically encoded and thus enabling non-invasive modulation of specific molecular events or biomolecular functions. Unlike light, ultrasound can reach into the deep brain tissue regions without surgical intervention which offers a non-invasive alternative for deep tissue modulation.
This white paper explores the limitations of current neural modulation techniques, while introducing sonogenetics as an alternative to these techniques and outlines its potential applications, challenges and future directions.
2. Problems and Strategic Solutions
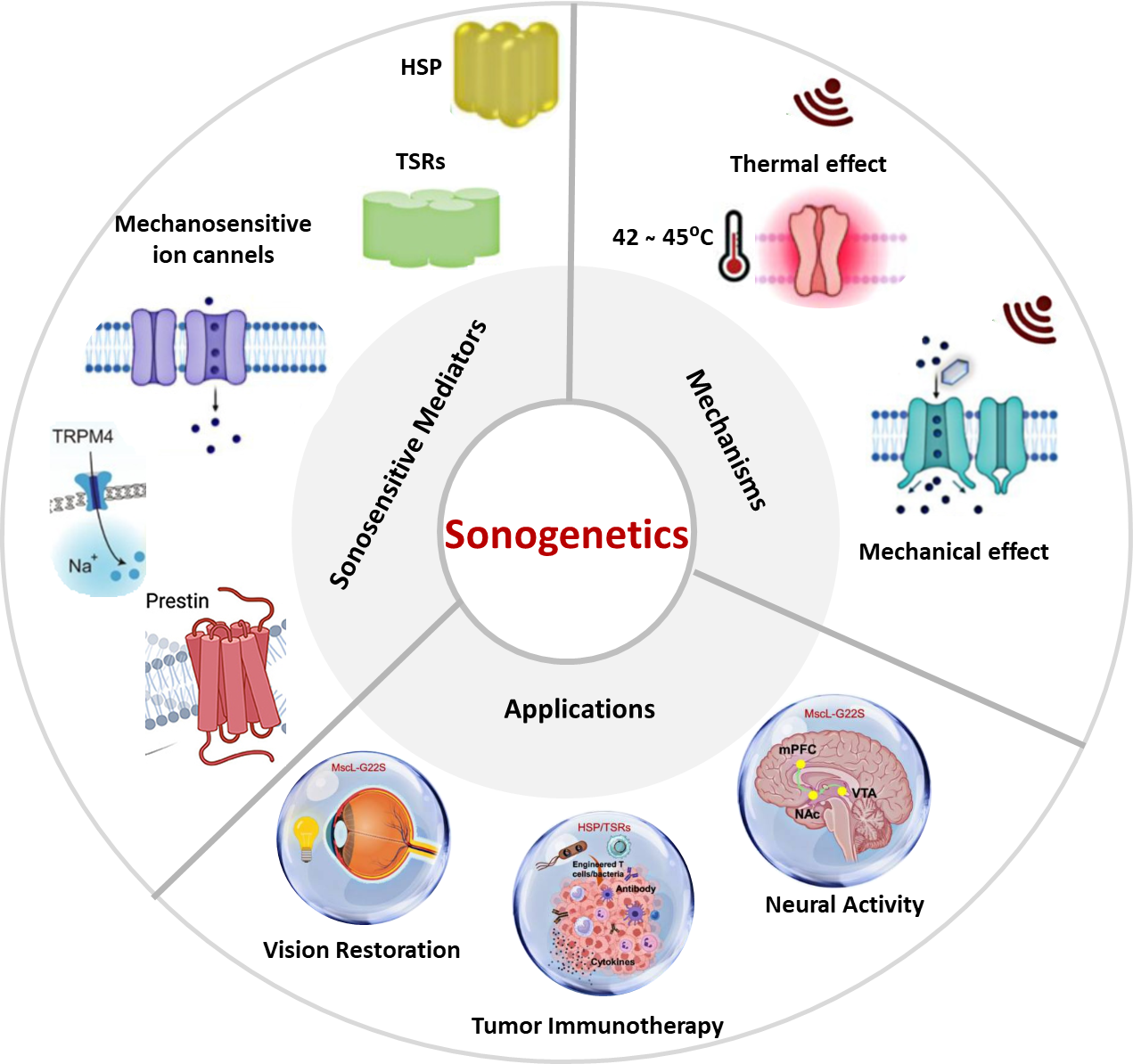
3. Sonosensitive Mediators
Sonosensitive mediators (SSMs) fall into two major categories that play a central role in linking sonogenetics to cellular function control, which has been identified across diverse organisms from bacteria to mammals.

3.1 Thermal Mediators
Heat Shock Protein (HSP) promoters:
HSP promoters are widely used by the body to synthesis proteins that helps stabilize cellular function during fever. In gene therapy, HSP70 promoters are valuable due to their ability in initiating and regulating transcription of therapeutic genes. HSP70 can enhance the gene expression thousand-fold under hyperthermic conditions. In a study, HSP70 promoter successfully controlled the transgene expression under localized hyperthermia both in vitro and in vivo. Magnetic resonance imaging (MRI) can be used to track ultrasound-induced hyperthermia, enabling precise control of therapeutic gene expression within defined temperature ranges. But, even short period of hyperthermia alone does not sustain gene activation and cellular function. To address this, the Cre-lox switch has been integrated into the HSP-driven system. In this design, transient Cre expression triggered by ultrasound removes a STOP cassette, shifting cells from ZsGreen to continuous production of the therapeutic protein. Traditionally, hyperthermia-inducible systems required cloning each gene under an HSP promoter, which complicated the regulation of multiple targets. The advanced approach now combines ultrasound hyperthermia with an enhanced CRISPR-Cas platform. Here, Cas proteins are controlled by a truncated HSP70 promoter that responds rapidly to heat, while customized guide RNA arrays enable simultaneous activation of one or more therapeutic genes, supporting multiplexed regulation.
Transient Receptor Potential (TRP) cationic channels:
Thermosensitive ion channels offer a powerful way to regulate biomolecular functions through ultrasound-induced hyperthermia, with particular attention on the TRP family. A study showed that the TRP vanilloid 1 (TRPV1) channel becomes active at temperatures above 42 °C, influencing neuronal synaptic activity. Importantly, this threshold lies only slightly above the normal body temperature of most mammals. As a result, TRPV1 remains inactive under typical physiological conditions but switches on when temperature is around 42 °C. In neurons expressing TRPV1, localized temperature elevations in the range of 38.5–39.7 °C have been reported to trigger activation. Even at low expression levels, TRPV1 is observed to produce strong responses, reducing the risk of toxicity often associated with introducing foreign proteins.
Temperature-Sensitive Repressors (TSR):
In 2017, researchers introduced orthogonal temperature-sensitive repressors (TSRs) capable of producing up to a hundred-fold change in gene expression in response to temperature changes. Current ultrasound-responsive TSRs are largely adapted from bacterial and phage transcriptional repressors, notably TlpA and TcI. In a study, the utility of TlpA36 engineered variant for precise spatiotemporal regulation of microbial therapeutics using ultrasound was demonstrated. In their study, E. coli engineered to express GFP under TlpA36 control was injected into the hindlimbs of nude mice. MRI-guided ultrasound was then applied to maintain a localized temperature of 41 °C for 45 minutes, leading to GFP expression specifically at the targeted site. This work highlighted the promise of sonogenetics as a tool for manipulating cellular functions in biomedical contexts.
3.2 Mechanical Mediators
Mechanosensitive ion channels (MscL) of large conductance:
MscL, first identified from the membrane of Escherichia coli, was the earliest mechanosensitive ion channel to be cloned. Members of MscL family are pore-forming proteins that convert mechanical stress on the cell membrane into electrical activity. These channels exhibit a high conductance of about 3 nanoSiemens (nS), allowing the passage of ions, water and even small proteins when open. Functionally, MscL serves as a stretch-activated valve, releasing cellular contents to osmotic shock.
Alterations to amino acid residues in the MscL channel can facilitate its ability to open or close in response to different physical stimuli. For example, the MscL G22C mutant located in the M1 transmembrane helix, when linked to light-sensitive compounds, allows reversible control of the channel pore's opening and closing. Similarly, coupling the G22C mutant with a pH-responsive molecule adjusts the pore's hydrophobicity by changing the surrounding pH. In another case, combining the MscL M42C mutant with the magnetic nanoparticle CoFe₂O₄ reduces the mechanical threshold needed for channel activation under a magnetic field.
Transient receptor potential (TRP) channel proteins:
TRP channel proteins form a large family of non-selective cation channels with weak voltage sensitivity, facilitating the inflow of cations. TRP channels are present in central and peripheral nervous system in humans. The TRP channel family includes seven subfamilies which are TRP canonical channels, TRP melastatin channels, TRP vanilloid channels, TRP melastatin channels, TRP mucolipin channels, TRP polycystin channels and TRP ankyrin channels. Each protein in the family has a six-transmembrane structure (TM1-TM6) where both N-terminal and C-terminal ends are located intracellularly. The pore that allows cation passage is formed by the fifth and sixth transmembrane domains. TRP channels exhibit diverse gating mechanisms, ranging from constitutive activity to regulation by voltage, mechanical stress, temperature, or chemical ligands.
In a study, researchers showed that TRP-4 ion channels that are key pore-forming components of mechanotransduction pathways, can be activated using ultrasound in the presence of microbubbles. However, this process depends on the presence of microbubbles (MBs), which restricts its practical use in mammalian systems.
Piezo ion channel:
Piezo proteins includes Piezo1 and Piezo2 in vertebrates, which are large membrane proteins that are built from three monomers arranged in a structure resembling a three-bladed propeller. They are the largest known class of transmembrane proteins as they span the membrane up to 114 times. The first 36 transmembrane segments form nine repeating units with each unit composed of four helices. The final two transmembrane domains are thought to create the pore through which ions pass.
These channels are uniquely activated by mechanical pressure. They open upon stimulation to allow cations to flow across the membrane. This property enables Piezo proteins to mediate cellular mechanotransduction which helps cells to adapt to their surrounding environment. Piezo1 selectively conducts Na⁺, K⁺ and Ca²⁺, while Piezo2 functions as a non-selective cation channel. Together, they play a central role in converting ultrasound-induced mechanical signals into downstream cellular responses.
Two-pore domain potassium (K2P) family:
The K2P family represents a class of potassium channels that are characterized by two pore domains, four transmembrane segments and extracellular caps. TREK-1, TREK-2 and TRAAK belongs to the K2P family and function as mechanosensitive channels that respond to physical force. They play important roles in regulating vital physiological functions and are widely expressed in the mammalian nervous system. Activation of these channels is closely influenced by the concentration of extracellular potassium (K⁺).
TRAAK channel activity depends on the tension within the lipid bilayer, allowing mechanosensitive gating and regulation of cellular function. It is specifically concentrated at the nodes of Ranvier, critical sites for action potential transmission in myelinated axons and contributes to the "leak" potassium current. Notably, ultrasonic energy can directly activate TRAAK through the cell membrane without requiring other cellular components. Even low-power ultrasound produces strong activation, with kinetics up to 20 times faster. Because of its low baseline open probability and relatively high conductance, TRAAK stands out as a promising ultrasound-responsive protein for advancing sonogenetics.
Prestin:
Prestin is a transmembrane protein in the cochlea that functions as a voltage-to-force motor. In outer hair cells (OHCs), changes in membrane potential drive its activity, contributing to the generation of auditory signals in mammals. Prestin is highly sensitive to sound frequencies below 20 kHz which allows it to play a crucial role in hearing. Its function relies on electromechanical signals transmitted from OHCs to the brain. Importantly, prestin is not an ion channel and does not participate in ion exchange across the membrane.
4. Process Workflow of Sonogenetics
4.1 Sound-sensitive proteins:
Identifying suitable sound-sensitive proteins for use in sonogenetics is a challenge. Sonogenetics relies on ultrasound waves to activate mechano-sensitive ion channel genes introduced into target cells and these channels respond to mechanical forces such as shear stress, stretching, pressure, or osmotic changes. The cell membrane deforms when mechanical force is applied, causing the channel helix to shift. This opens the pore, allowing ions and small molecules to pass through.
4.2 Gene delivery and expression:
Genes that encode ultrasound-sensitive proteins can be introduced into target cells through gene delivery techniques. This is typically achieved using viral or non-viral vectors and in some cases by generating transgenic lines.
4.3 Ultrasound exposure:
Ultrasound is an acoustic wave with a frequency above 20,000 Hz. In non‑invasive surgical method to thermally ablate tissues, focused high‑intensity ultrasound is used. The effectiveness of focused ultrasound depends on delivering the right amount of energy within a short time. The key parameters such as intensity, frequency, duration, duty cycle and pulse repetition frequency are carefully adjusted to optimize results.
- Intensity: high intensity focused ultrasound (100 W/cm²–10 kW/cm²) for surgery; low intensity focused ultrasound (<3 W/cm²) for modulation; >100 mW/cm² disrupts brain activity.
- Frequency: High frequency (1–20 MHz) for diagnosis, Medium (0.7–3 MHz) for therapy, Low (20–200 kHz) for industry; lower frequency results in deeper penetration.
- Duration: Short-term low intensity focused ultrasound stimulates neurons; long-term (>10 s) inhibits activity.
- PRF: >500 Hz enhances neural activity (evoked EEG).
- Duty Cycle: Continuous = 100%; pulsed (<100%) effective for neural activation.
4.4 Readout:
The effects of stimulating ultrasound-sensitive proteins need to be assessed in cells, tissues, or whole organisms. This can be done using electrodes and arrays to measure changes in membrane voltage by tracking calcium signals after ultrasound exposure. Various biosensors can be used to help capture different cellular responses such as dyes and genetically encoded indicators. And finally, studying cell behavior provides insight into how ultrasound can modulate activity in vivo.
5. Applications: Visual Restoration, Immunotherapy and Neuromodulation
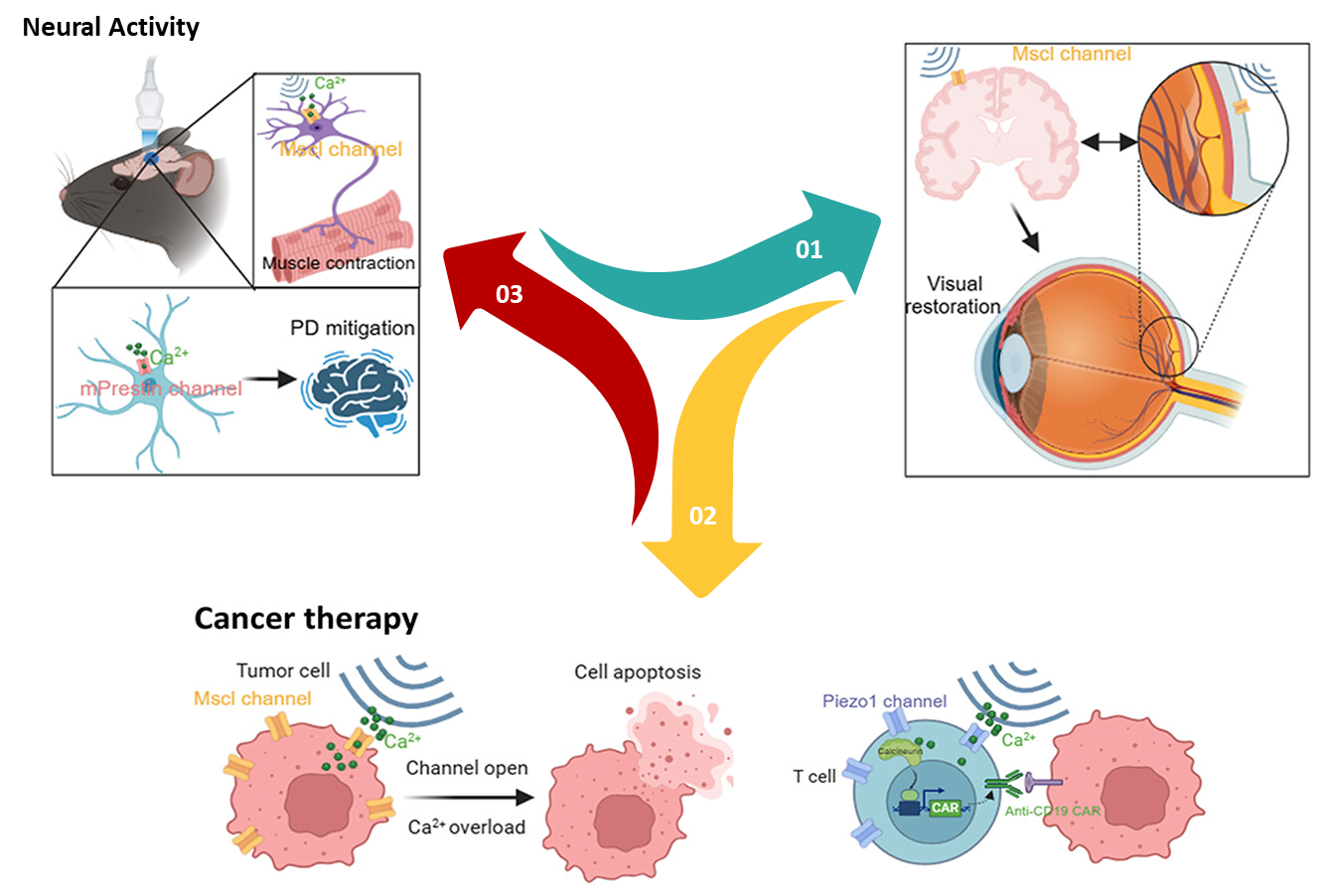
5.1 Visual Restoration
Retinitis pigmentosa is a leading cause of vision loss caused by the progressive breakdown of light-sensitive cells in the retina, resulting in profound sensory impairment. In 2023, a study revealed that combining MscL-G22S ion channels with high-frequency ultrasound stimulation leads to activation of retinal or cortical neurons with exceptional spatiotemporal precision and acoustic energy deposition for vision restoration.
MscL-G22S was expressed in rat retinal ganglion cells (RGCs) and ultrasound stimulation at 15 MHz produced strong and sustained ON-type spiking activity, with most RGCs responding at ultra-short latencies. Cortical neurons expressing MscL-G22S exhibited millisecond-scale responses and achieved spatial resolution of approximately 400 µm in the x-y plane. Behavioral experiments further supported these findings: mice trained to associate visible-light stimulation of one eye with a water reward showed significantly higher anticipatory licking success after cortical sonogenetic stimulation compared to non-transfected controls. This suggests that the stimulation enabled a degree of light perception. Therefore, sonogenetics has been reported as a significant advancement in published studies in developing high-resolution strategies for visual restoration at the cortical level.
5.2 Tumor Immunotherapy
Disruption in the balance of ion channel proteins and transporters can cause cells to swell and undergo oncosis, opening up a potential strategy for cancer treatment. Mechanosensitive channel proteins found in bacterial membranes can be engineered or chemically modified to respond to external stimuli such as ultrasound. By altering calcium homeostasis, these channels can drive cells toward programmed death. A logic AND-gated sonogenetic nanosystem was developed based on this principle using cationic nanoliposomes. Under continuous ultrasound exposure the MscL channels remain open allowing excessive calcium entry that activates calcium-dependent apoptotic pathways thereby leading to mitochondrial dysfunction, suppressing tumor growth.
Mechanosensitive ion channel Piezo1 is highly expressed in pancreatic ductal adenocarcinoma (PDAC) tissues and cell lines, where its presence correlates with advanced disease stage and reduced disease-free survival. Researchers employed microbubbles to enhance ultrasound signals activating Piezo1 that triggered calcium influx, disrupted mitochondrial function and ultimately apoptosis in pancreatic cancer cells.
5.3 Neuromodulation and Nervous System Disease
Optogenetic neuromodulation is well known for its high spatial and temporal resolution as well as cell-type specificity, but it has limitations such as shallow penetration of light, especially in deeper brain regions. In contrast, sonogenetics uses ultrasound to achieve noninvasive, deep tissue penetration while still maintaining precise targeting of specific cells. Most studies in sonogenetic neuromodulation have reported calcium influx and the generation of action potentials in neurons. In addition, electromyogram recordings have captured muscle responses following transcranial ultrasound stimulation of brain regions, demonstrating modulation of motor-related neural activity.
Research has shown that when exposed to low-intensity ultrasound, MscL-G22S-expressing neurons exhibit pronounced calcium influx and generate measurable electromyogram responses in brain cell populations of living animals. In experiments where neurons within the right dorsomedial striatum (DMS) of mice were engineered to express MscL-G22S, ultrasound stimulation produced markedly stronger neuronal activation compared to non-expressing controls. This heightened activity was confined to the right DMS and did not spread to other brain regions, therefore showing the potential of this approach for precise, non-invasive and repeatable modulation of deep brain circuits.
6. Challenges in Advancing Sonogenetics
Identifying suitable ultrasound mediators
Sonogenetics requires finding the right mechanosensitive (MS) ion channels. The current strategies includes top-down screening that relies on calcium imaging, but is limited by sensitivity and bottom-up channel engineering which is effective but labor-intensive.
Safety and technical concerns
Genetic engineering with viral vectors raises risks of immune responses and off-target activation of endogenous MS ion channels reduces efficiency. Ultrasound can activate the cochlear pathway and can exhibit secondary mechanical effects similar to that evoked by audible sound in animal models. Further, high-intensity focused ultrasound (HIFU) can cause irreversible tissue damage.
Designing ultrasound-sensitive proteins
There is still a need for designing proteins responsive to low-intensity ultrasound to improve safety and minimize tissue disruption. It requires systematic high-throughput screening to identify candidates with structural and functional compatibility. To evaluate efficacy and side effects in-depth testing in cellular and in vivo environments is essential.
Limitations in ultrasound devices and procedures
The need for high-resolution focusing and precise control of ultrasound to ensure uniform mechanical and thermal effects. Advancements in transducer technology and imaging are required to achieve accurate targeting. Neuromodulation requires precise focusing when targeting the complex structures of the brain or accessing its deeper regions.
7. Future Directions
Sonogentics has been observed as a promising tools for non-invasive modulation and therapy. While showing promising applications there are significant refinement still required before it makes major impact in medicine. Early studies highlights the potential of ultrasound in neurostimulation and treating neurological disorders, however applications in modeling neuropathologies remains limited. Expanding this work in future could enable the creation of animal models that can better replicate human neurological diseases, allowing researchers to probe signaling pathways, manipulate specific cell types and control disease progression with greater precision.
Accurate disease modeling is vital for uncovering underlying mechanisms, tracking progression and testing therapies. Beyond current focus areas there are unexplored conditions such as neurodegenerative and psychiatric disorders which include memory loss, depression and anxiety, are being explored as potential areas of investigation for advancing mental health research.
At present, most efforts in sono-synthetic biology remain at the laboratory stage, centered on developing sonogenetic switches. So, rigorous safety validation, deeper mechanistic insights and careful clinical translation will be essential to unlock the therapeutic potential of these technologies.
8. Conclusion
Sonogenetics combines ultrasound and synthetic biology to enable precise and non-invasive control of cellular activity. Early studies show promise in areas like vision restoration, tumor therapy and neuromodulation, however these applications are still at an early stages. There are also some key challenges such as identifying reliable mediators, ensuring safety, designing proteins responsive to low-intensity ultrasound and improving device precision.
Despite these challenges, the published studies suggest that sonogenetics is a promising platform for non-invasive cellular modulation and it could transform disease modelling while opening new avenues for treating neurological and psychiatric disorders and help in expanding non-invasive medicine. Continued innovation, safety validation and careful clinical translation will be essential to realize its full impact.



-Sensors-Next-Generation-Flexible-Devices/Wrinkled-Silver-Nanowire-AgNW-Sensors-Next-Generation-Flexible-Devices.png)




