GLP-1 Receptor Agonists in Neurodegenerative Diseases: Opportunities, Limitations & the Road Ahead
Executive Summary
Glucagon-like peptide-1 (GLP-1) is a gut-derived incretin hormone best known for its role in glucose homeostasis, appetite regulation, and energy balance. GLP-1 receptor agonists (GLP-1RAs) are widely approved for the treatment of type 2 diabetes mellitus (T2DM) and obesity. Over the past decade, growing evidence has demonstrated that GLP-1 receptors are also expressed in multiple regions of the central nervous system (CNS), prompting investigation into their potential role in neurodegenerative diseases.
Preclinical studies consistently demonstrate neuroprotective effects of GLP-1RAs in models of Alzheimer's disease (AD), Parkinson's disease (PD), multiple sclerosis (MS) and amyotrophic lateral sclerosis (ALS). These effects include reduced neuroinflammation, improved mitochondrial function, enhanced neuronal survival, and modulation of protein aggregation pathways. However, translation into clinical efficacy has been inconsistent, with human trials yielding mixed or modest outcomes.
Key challenges include limited and variable blood–brain barrier (BBB) penetration, uncertainty regarding effective CNS target engagement, and tolerability concerns, particularly gastrointestinal adverse effects in elderly and frail populations. Emerging strategies such as nanoparticle-based delivery systems, improved biomarkers, and artificial intelligence-driven patient stratification may help address these limitations and refine the therapeutic positioning of GLP-1RAs in neurodegenerative disease.
1. Introduction
Glucagon-like peptide-1 (GLP-1) is produced through post-translational processing of the proglucagon gene and is secreted primarily by enteroendocrine L-cells in the small intestine following nutrient intake. In addition to its peripheral secretion, GLP-1 is also synthesized in discrete regions of the brain and hypothalamus that regulate appetite, energy expenditure, and glucose metabolism.
Physiologically, GLP-1 secretion increases two to three folds following food intake, enhancing insulin secretion through the incretin effect while suppressing glucagon release. Native GLP-1 exists predominantly as GLP-1 (7-36) amide and GLP-1 (7-37), but it is rapidly degraded in circulation by dipeptidyl peptidase-4 (DPP-4) and neprilysin, resulting in a short half-life of only a few minutes.
GLP-1 exerts its biological activity by binding to the GLP-1 receptor (GLP-1R), a class B G-protein-coupled receptor expressed in pancreatic islets, the gastrointestinal tract, cardiovascular tissues, and the CNS. The development of long-acting GLP-1 receptor agonists resistant to enzymatic degradation has enabled widespread clinical use in metabolic diseases and opened new avenues for neurological applications.
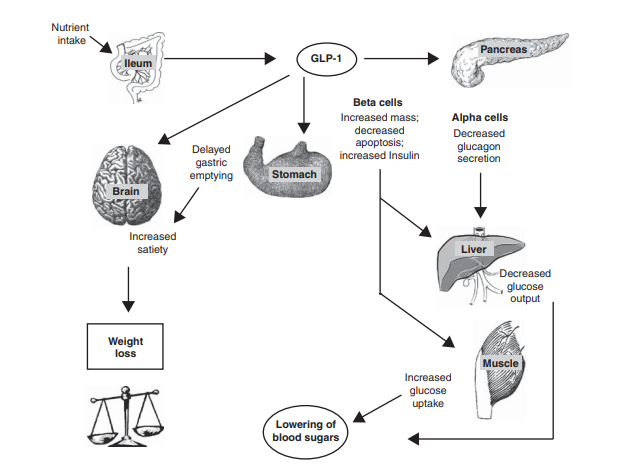
Fig. 1 Sources and uses of GLP-1 (Source: Alan J Garber, et al. 2011)
2. Mechanism of Action of GLP-1 Signaling
GLP-1 signaling is initiated upon ligand binding to GLP-1R, leading to activation of adenylate cyclase and an increase in intracellular cyclic AMP (cAMP). Elevated cAMP activates protein kinase A (PKA) and exchange proteins directly activated by cAMP (EPAC), resulting in downstream effects on insulin secretion, cell survival, and metabolic regulation.
In pancreatic β-cells, GLP-1 signaling enhances glucose-dependent insulin secretion, promotes β-cell survival and proliferation through PI3K/Akt pathways, and improves glucose sensitivity. In α-cells, GLP-1 indirectly suppresses glucagon secretion through paracrine effects mediated by insulin and somatostatin released from β- and δ-cells, respectively.
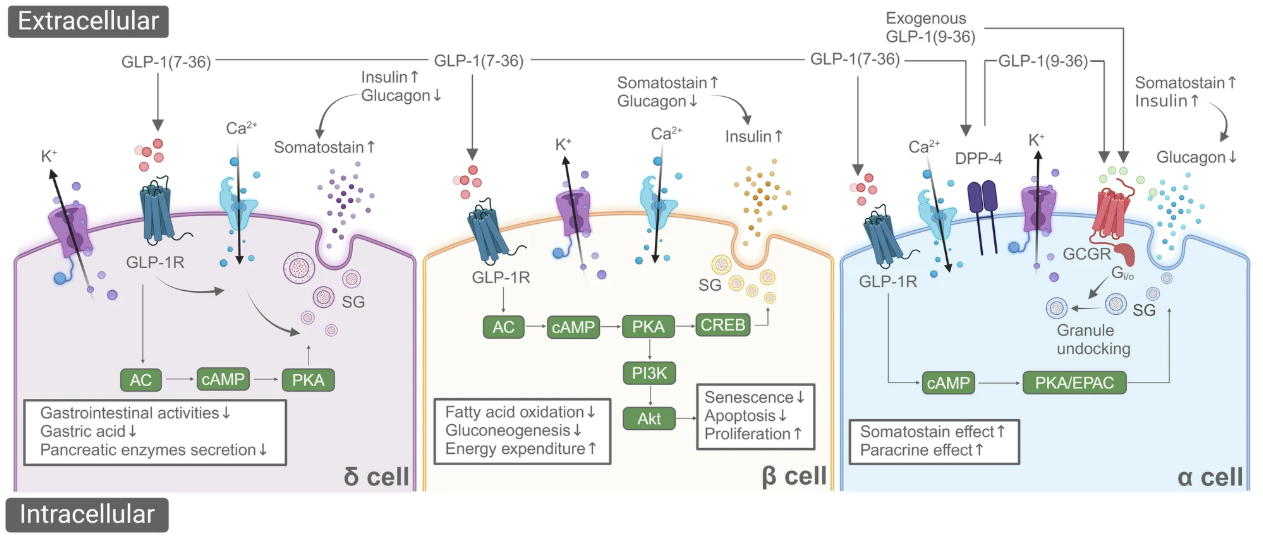
Fig. 2 Mechanism of GLP-1 to regulate blood glucose (Source: Zhikai Zheng, et al. 2024)
Beyond the pancreas, GLP-1 signaling influences gastric emptying, appetite regulation, energy expenditure, and lipid metabolism. Importantly, GLP-1 receptors expressed in the CNS mediate neurotrophic, anti-inflammatory, and mitochondrial protective effects that form the mechanistic basis for exploration in neurodegenerative disorders.
3. Clinical Use of GLP-1RAs in Metabolic Disease
GLP-1 receptor agonists have an established safety and efficacy profile in metabolic indications. Approved agents include exenatide, liraglutide, dulaglutide, lixisenatide, semaglutide, and the dual GLP-1/GIP agonist tirzepatide. These therapies improve glycemic control, promote weight loss, and reduce cardiovascular risk in patients with T2DM and obesity.
The metabolic benefits of GLP-1RAs, including improved insulin sensitivity, reduced systemic inflammation, and enhanced mitochondrial function, are increasingly recognized as potentially relevant to neurodegenerative disease pathology, where metabolic dysfunction and neuroinflammation play contributory roles.
4. GLP-1RAs and Neurodegenerative Diseases
Glucagon-like peptide-1 receptor agonists (GLP-1RAs), originally developed for the treatment of type 2 diabetes and obesity, have recently gained attention in the field of neurodegenerative diseases. Beyond their metabolic benefits, GLP-1RAs exhibit neuroprotective properties such as reducing neuroinflammation, enhancing mitochondrial function, and promoting neuronal survival making them a good option for treatment of diseases such as Alzheimer's disease, Parkinson's disease, Multiple Sclerosis, and Amyotrophic Lateral Sclerosis.
4.1. Alzheimer's disease:
In preclinical models of Alzheimer's disease, GLP-1RAs such as liraglutide , semaglutide, and exenatide reduce amyloid-β accumulation, inhibit tau hyperphosphorylation, and attenuate neuroinflammatory responses. These agents promote synaptic preservation, enhance neurogenesis, and improve cognitive performance in animal studies.
Early clinical studies have shown stabilization of cerebral glucose metabolism but limited cognitive benefit. Large phase III trials, including EVOKE and EVOKE+, are currently evaluating oral semaglutide in early symptomatic AD, with a focus on biomarker modulation and disease progression.
Fig.3 Types of neurodegenerative diseases
4.2. Parkinson's disease:
GLP-1RAs target several key pathological mechanisms in Parkinson's disease, including mitochondrial dysfunction, neuroinflammation, and dopaminergic neuron loss. Preclinical studies demonstrate activation of PI3K/Akt signaling, reduced apoptosis, and improved neuronal survival.
While early trials with exenatide showed improvements in motor scores, subsequent phase III studies failed to demonstrate disease-modifying effects. More recently, lixisenatide demonstrated stabilization of motor symptoms in early PD, suggesting potential benefit in selected patient populations.
4.3. Multiple Sclerosis:
In animal models of multiple sclerosis, GLP-1RAs delay disease onset, reduce severity, and promote remyelination. These effects are mediated through modulation of microglial and macrophage activation, reduction of pro-inflammatory cytokines, and enhanced oligodendrocyte precursor differentiation.
Clinical data remain limited but suggests acceptable safety and tolerability. Long-term efficacy and optimal patient selection require further investigation.
4.4. Amyotrophic Lateral Sclerosis:
Evidence supporting the use of GLP-1RAs in ALS is currently limited and primarily preclinical. Some studies suggest potential antioxidant and neuroprotective effects, but results are inconsistent across models. Well-designed, disease-specific clinical trials are required before therapeutic relevance can be established.
5. Clinical Trial Landscape
Across neurodegenerative indications, clinical trials of GLP-1RAs have demonstrated biological activity but variable clinical efficacy. Challenges include heterogeneity in disease stage, insufficient CNS exposure, and lack of sensitive biomarkers to detect early therapeutic effects.
Recent advances in PET imaging, fluid biomarkers, and digital endpoints are improving trial design and may enable better assessment of CNS target engagement and treatment response.
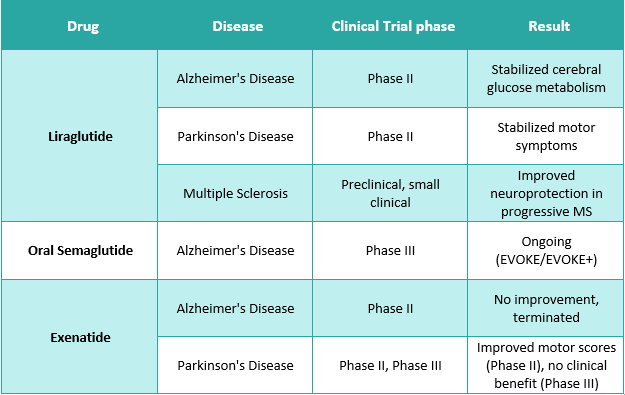
Table 1. Summary of drugs being investigated for Alzheimer's disease, Parkinson's disease, and Multiple Sclerosis along with their clinical trial phase and outcome.
6. Key Challenges in CNS Translation
Despite their many benefits, challenges like limited blood-brain barrier penetration, tolerability, adherence, and safety of GLP-1 still remains for treatment of neurodegenerative diseases such as Alzheimer's disease, Parkinson's disease, etc.
Blood–Brain Barrier Penetration
Although GLP-1RAs can access the CNS, their penetration across the BBB is limited and variable. Large, hydrophilic peptide structures restrict efficient transport, resulting in low cerebrospinal fluid concentrations even after prolonged dosing.
Tolerability, Adherence, & Safety
Gastrointestinal adverse effects, including nausea, vomiting, diarrhea, and weight loss, are common, particularly during dose escalation. These effects may be more pronounced in elderly patients with neurodegenerative diseases and can limit long-term adherence.
Clinical Heterogeneity
Neurodegenerative diseases are biologically heterogeneous, and metabolic modulation may only benefit specific patient subgroups or early disease stages.
7. Future Directions
Overcoming current limitations will require a combination of technological and clinical innovation. Nanoparticle-based delivery systems offer potential to enhance BBB penetration and CNS exposure while reducing systemic side effects. Targeted formulations may enable lower dosing and improved tolerability.
Artificial intelligence and data-driven tools can support patient stratification, identify predictive biomarkers, optimize dosing strategies, and inform combination therapy approaches. Future clinical trials are likely to focus on early-stage or at-risk populations, where disease modification may be more achievable.
From an innovation and IP perspective, current research and development efforts increasingly emphasize CNS-targeted delivery platforms, optimized formulations, patient stratification biomarkers, and combination regimens, as reflected in ongoing academic and translational studies. These areas represent key opportunities for next-generation development and strategic positioning.
8. Conclusion
GLP-1 receptor agonists represent a promising but still evolving therapeutic strategy in neurodegenerative diseases. While preclinical evidence strongly supports neuroprotective mechanisms, clinical translation has proven challenging. Current evidence indicates that GLP-1RAs are being evaluated across multiple neurodegenerative indications, with observed effects primarily associated with metabolic regulation and modulation of neuroinflammatory pathways in specific clinical settings.
Continued innovation in delivery technologies, biomarker development, and patient selection will be critical to realizing the full potential of GLP-1-based therapies in CNS disorders.



-Sensors-Next-Generation-Flexible-Devices/Wrinkled-Silver-Nanowire-AgNW-Sensors-Next-Generation-Flexible-Devices.png)





