1. Introduction
Antibody-drug conjugates (ADCs) are widely used for cancer therapies due to their potential in the delivery of cytotoxins to targeted cancer cells. The first generation of ADC, represented by gemtuzumab ozogamicin, employed antibodies conjugated randomly to cytotoxic agents such as calicheamicin. However, these findings suffered due to unstable linkers, inconsistent drug to antibody ratios (DARs), and restricted control over payload delivery, which eventually led to off-target toxicity. The second generation ADCs used antibodies, cleavable linkers, and modified conjugation sites which improved DAR consistency and further enabled the use of potent microtubule inhibitors like MMAE and DM1. The third generation ADCs have emphasized on site specific conjugation strategies, use of tumor responsive linkers, and incorporation of next-generation payloads such as topoisomerase I inhibitors e.g., DXd and SN 38.
Despite all these innovations, ADCs still remain burdened by considerable clinical obstacles. Cancer cells can adapt and develop resistance to drugs, which is still a challenge in effective cancer treatment. Cancer cells develop drug resistance through mechanisms including metabolic irregularities, target antigen loss or mutation, impaired internalization, lysosomal dysfunction, and upregulation of efflux pumps. Recent progress in conjugation methods has given rise to dual‑payload ADCs for the use in comination therapy. Unlike traditional ADCs, which were designed to deliver a single payload to an antigen, dual-payload ADCs are engineered by conjugating two cytotoxic agents onto a single antibody.
2. Problems and Pathways Forward
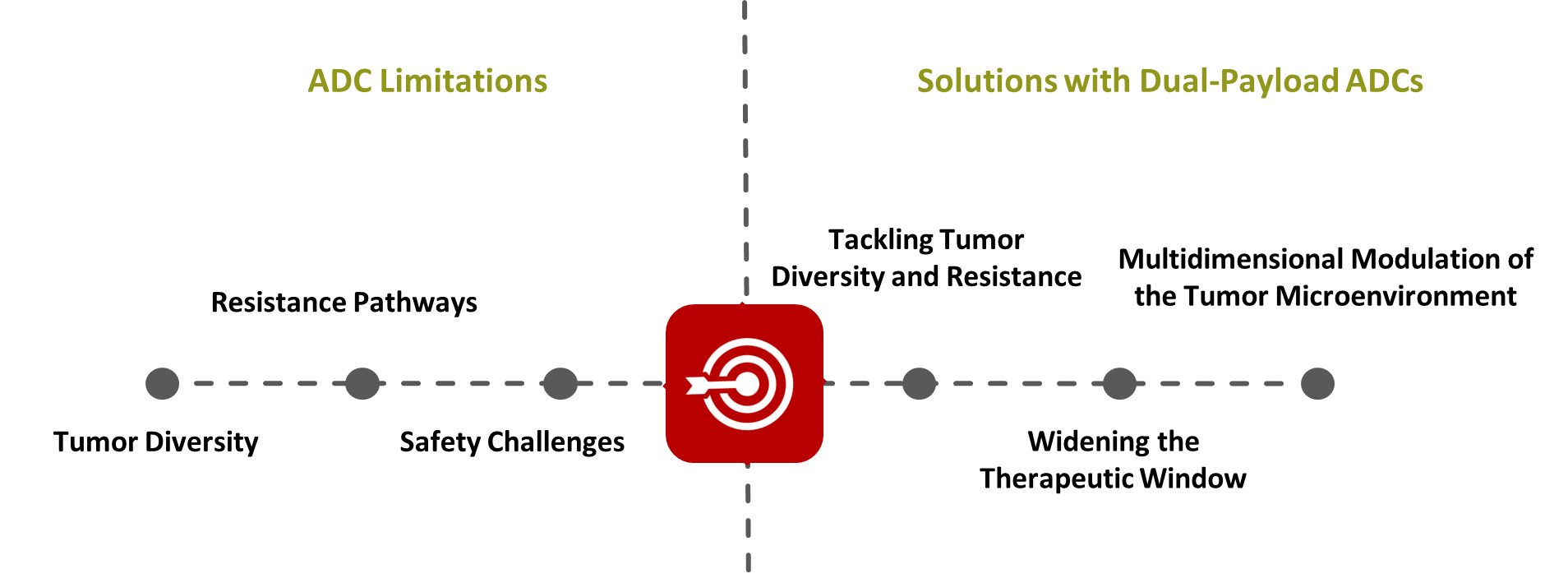
2.1 ADC Limitations: The Roadblocks Ahead
Tumor Diversity
Tumors evolve continuously and develop multiple subclones with distinct biological characteristics, a phenomenon known as intra-tumor clonal evolution. These subpopulations often exhibit varying sensitivities to therapeutic agents. For example, HER2 expression in breast cancer can vary by 2–5 log orders between different subtypes, making it difficult to standardize a single ADC therapy for all patients.
Even within the same patient, different regions of a tumor—or tumors across different patients—may express target antigens at highly variable levels. As a result, single-payload ADCs may fail to eliminate all malignant clones, increasing the risk of disease relapse. This highlights the need for therapeutic strategies capable of adapting to dynamic tumor evolution.
Resistance Pathways
Cancer cells can develop resistance to ADCs through multiple biological mechanisms:
- Antigen downregulation: Cancer cells reduce the number of surface antigens, limiting ADC binding and payload delivery.
- Epitope masking: Structural alterations in target antigens prevent effective antibody recognition and binding.
- Lysosomal dysfunction: Impaired lysosomal acidification disrupts payload release after endocytosis, rendering ADCs ineffective.
- Drug efflux mechanisms: Activation of transporters such as MDR1/P-glycoprotein expels cytotoxic payloads before they can exert therapeutic action.
Safety Challenges
Increasing payload potency can help overcome resistance, but it also raises the risk of off-target toxicity to healthy tissues. Instability in covalent linker chemistry can result in premature payload release, leading to hematotoxicity and damage to vital organs.
Studies indicate that nearly 40% of ADC-related adverse effects are attributed to poor linker stability, underscoring the urgent need for more precise and reliable drug-release mechanisms.
2.2 Therapeutic Solutions with Dual-Payload ADCs
Tackling Tumor Diversity and Resistance
Tumor heterogeneity means that different cancer cells within the same tumor may respond to different classes of cytotoxic agents. Dual-payload ADCs increase the probability of eliminating diverse malignant populations by delivering two complementary payloads simultaneously.
By integrating two drugs with distinct mechanisms of action into a single ADC, dual-payload therapies can target multiple intracellular pathways. This dual mechanism enables continued tumor killing even if cancer cells develop resistance to one of the payloads.
Widening the Therapeutic Window
In dual-payload ADC therapy, both cytotoxic agents are delivered using a single antibody-targeting system, which limits exposure to healthy tissues compared to administering two separate drugs.
Careful optimization of the payload ratio allows maintenance of therapeutic efficacy while minimizing the toxicity commonly associated with high-dose single-agent treatments.
Multidimensional Modulation of the Tumor Microenvironment
Certain dual-payload ADCs are designed to combine cytotoxic agents with immune-modulating payloads. This approach not only induces direct tumor cell death but also stimulates immune activity within the tumor microenvironment.
The resulting immunogenic cell death enhances antitumor immune responses and enables more effective regulation of the tumor ecosystem, offering benefits beyond those achievable with single-payload ADCs.
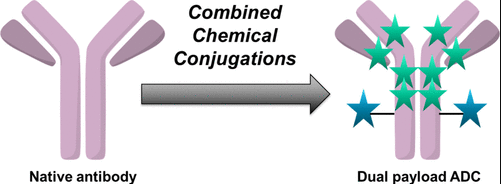
3. Techniques for Dual-Payload ADC Construction
The strategies for generating homogeneous dual payload ADCs can be classified into three main categories, based on the methods of introduction of orthogonality.
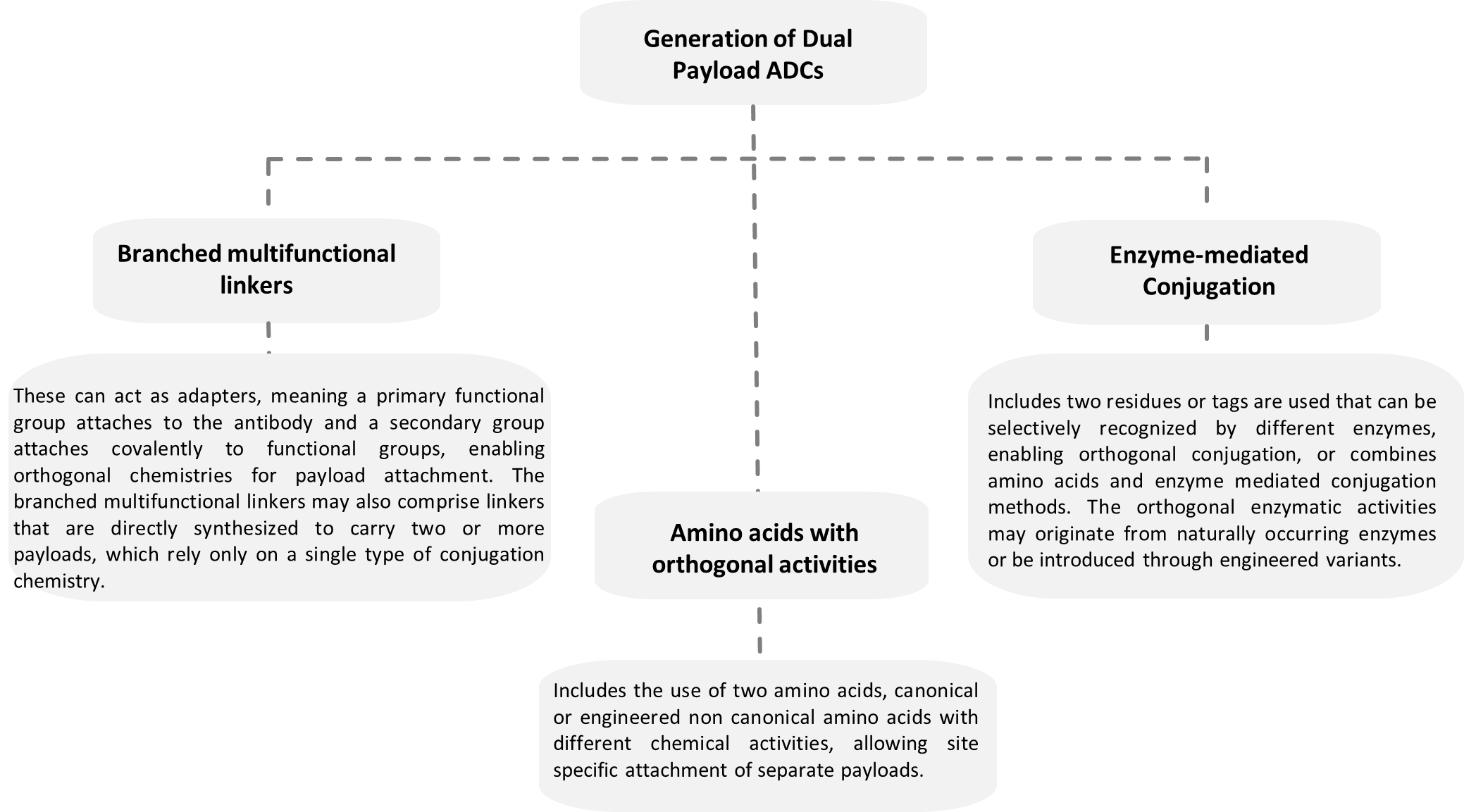
3.1 Branched Multifunctional Linkers
Multifunctional linkers contain three or more branches, where one branch is typically used to attach the antibody or protein scaffold, while the remaining branches are designed to connect reactive functional groups or directly attach multiple payloads. This architecture enables the conjugation of two distinct cytotoxic agents through orthogonal chemistries while preserving antibody stability.
In a notable study, researchers at Seagen developed an adaptor incorporating two cysteine residues protected by different chemical groups to enable selective and sequential conjugation. One cysteine was masked with a reducible disulfide group, removable using tris(2-carboxyethyl)phosphine (TCEP), while the second cysteine was protected by an acetamidomethyl group, stable under TCEP conditions but removable under mild aqueous treatment with mercury acetate.
Using this orthogonal protection strategy, the adaptor was first conjugated to the interchain thiols of a CD30-targeting antibody via maleimide chemistry, enabling precise attachment of distinct linker–payload combinations.
3.2 Amino Acids with Orthogonal Reactivity
Many site-specific conjugation strategies rely on amino acids with unique chemical reactivity. Among the 20 naturally occurring amino acids, cysteine is most frequently used due to its strong nucleophilic nature. Lysine, the second most nucleophilic residue, has also become an important target for controlled conjugation approaches.
Lysine-directed conjugation strategies are actively explored and can be achieved by modifying the local protein environment or carefully selecting compatible conjugation partners to ensure specificity.
Canonical Amino Acids
Antibody–drug conjugates developed using engineered cysteine residues, known as THIOMAB ADCs (TDCs), were the first homogeneous ADCs shown to deliver an improved therapeutic index. This improvement was largely attributed to enhanced linker–payload stability achieved through precise site selection, which significantly reduced systemic toxicity.
The flexibility of engineered cysteines has led to their widespread adoption, and the addition of extra conjugation handles to antibodies already containing engineered cysteines has become a standard strategy for constructing homogeneous dual-payload ADCs.
A HER2-targeting scFv-Fc construct demonstrated the combination of selenocysteine with engineered cysteine to enable homogeneous dual-payload conjugation. In this approach, the THIOSELENOMAB conjugate underwent mild reduction to expose both residues. An iodoacetamide linker was coupled to selenocysteine under acidic conditions, followed by cysteine modification at basic pH using a methylsulfone phenyloxadiazole (ODA) linker, which offers improved stability over conventional maleimide linkers.
This strategy was later extended to conjugate both PNU and MMAE payloads onto a HER2-targeting antibody. The resulting dual-payload ADC showed potency comparable to the PNU-only ADC, indicating that cytotoxic activity was primarily driven by the PNU component. Morphological analysis confirmed the presence of dual mechanisms of action, including DNA damage and tubulin inhibition.
Non-Canonical Amino Acids (ncAAs)
The incorporation of non-canonical amino acids (ncAAs) has emerged as a powerful approach for developing second-generation ADCs that are both highly uniform and stable. Amber stop codon suppression is commonly used to introduce ncAAs, requiring an orthogonal aminoacyl-tRNA synthetase (aaRS) and its matching tRNA.
Unlike cysteine-based or enzyme-mediated approaches, ncAA-based conjugation enables direct and highly specific payload attachment without the need for reduction steps or multi-stage reactions. This simplifies development, improves manufacturability, and ensures consistent product quality.
In one study, a tryptophanyl aaRS/tRNA pair from E. coli was shown to function as an opal codon (TGA) suppressor. Combined with an orthogonal leucyl aaRS/tRNA amber suppressor, researchers successfully incorporated 5-hydroxytryptophan (5-HTP) and the azide-containing amino acid LCA into positions 121 and 198 of the trastuzumab heavy chain.
This enabled the generation of a dual-payload ADC through a one-pot reaction, in which diazo-MMAF and DBCO–PNU159682 were conjugated, demonstrating the efficiency and scalability of ncAA-based dual-payload strategies.
3.3 Enzyme-Mediated Conjugation
Enzyme-mediated conjugation strategies often require the placement of conjugation tags at the protein’s C-terminus, which can restrict labeling efficiency, reduce site-selection flexibility, and limit fine-tuning of dual-payload ratios. These approaches typically involve two-step reactions—adapter attachment followed by payload conjugation—potentially reducing overall yield.
Several studies have explored combining orthogonal enzyme-mediated strategies to generate homogeneous dual-payload ADCs, using either natural enzymes or engineered variants.
Natural Enzymes
To generate homogeneous dual-payload ADCs, researchers combined microbial transglutaminase (mTG) with lipoate acid ligase (LpIA). LpIA specifically recognizes a 13-residue sequence known as the LpIA acceptor peptide (LAP).
In this study, a lipoate acid trans-cyclooctyne (TCO) linker was first attached to the LAP tag at the C-terminus of the light chain, achieving conjugation efficiency greater than 85%. Subsequently, mTG was used to install a PEG3-azide linker at the Q295 site of the trastuzumab heavy chain.
After desalting, two fluorescent payloads were introduced: rhodamine, linked via a matrix metalloproteinase-2 cleavable extracellular linker, and fluorescein, attached through a cathepsin B-cleavable intracellular linker. Each payload was site-specifically conjugated to its respective adaptor.
Engineered Enzymes
A key limitation of natural sortase enzymes is their slow catalytic rate. To overcome this, researchers used yeast display technology to evolve a variant known as eSrtA, achieving a 140-fold increase in catalytic activity compared to the wild-type enzyme.
Two orthogonal sortase A variants, eSrtA (2A–9) and eSrtA (4S–9), were engineered to selectively recognize the substrates LAXTG and LPXSG, respectively. This enabled high activity and specificity through enhanced substrate recognition.
The first demonstration of homogeneous dual-payload conjugation using these variants involved attaching a PEG-modified polyglycine and an Alexa Fluor 750-tagged LAETG peptide to fibroblast growth factor 1 (FGF1). This method was later extended to conjugate two payloads to the C-termini of Fab heavy and light chains, achieving near-quantitative conversion with rapid kinetics.
4. Balancing Stability and Release: Linker Technologies
The therapeutic effectiveness of dual-payload ADCs is largely determined by the linker systems that connect cytotoxic drugs to the antibody. These linkers must remain highly stable in circulation to prevent premature drug release and systemic toxicity, while also being capable of efficient cleavage within tumor cells to ensure precise payload delivery.
Cleavable Linkers
Cleavable linkers are widely used in ADC design because they respond to tumor-specific physiological conditions, enabling selective intracellular drug release. Common triggering mechanisms include:
- Acidic pH
- Elevated intracellular glutathione levels
- Lysosomal enzymes
A key challenge in dual-payload ADC design is coordinating the release of both drugs, which may occur simultaneously or sequentially depending on therapeutic objectives. Disulfide linkers, for example, exploit high intracellular glutathione concentrations to undergo reductive cleavage, releasing thiol-containing cytotoxic agents.
Peptide-based linkers, such as valine–citrulline, are substrates for cathepsin B, a lysosomal protease commonly overexpressed in tumor cells. These linkers enable highly selective payload release within cancer cells. By combining multiple cleavable linker types, researchers can fine-tune the timing and location of drug activation, maximizing tumor cell killing while minimizing systemic exposure.
Non-Cleavable Linkers
Non-cleavable linkers offer an alternative strategy when maximum circulatory stability is required. Unlike cleavable systems, these linkers do not respond to chemical or enzymatic triggers. Instead, payload release occurs only after the ADC is internalized and degraded within lysosomes.
This approach is particularly effective when one payload benefits from controlled intracellular release while another payload exerts its effect following antibody breakdown. For example, pairing a non-cleavable maleimide linker with a microtubule inhibitor and a cleavable hydrazone linker with a DNA-damaging agent can create a staggered release profile that reduces off-target toxicity while enhancing tumor cell destruction.
Recent advances include the development of bifunctional linkers capable of attaching two distinct payloads to the same antibody position. This innovation improves drug-to-antibody ratio (DAR) consistency, reduces product heterogeneity, and enhances the predictability of ADC behavior in vivo.
Additionally, linker hydrophilicity plays a crucial role in ADC performance. Hydrophilic modifications such as PEGylation improve solubility, reduce aggregation, slow systemic clearance, and enhance tumor penetration, further optimizing therapeutic outcomes.
5. Key Challenges in Dual-Payload ADC Development
Although dual payload ADCs hold considerable therapeutic promise, their development is hindered by numerous challenges.

- 5.1 Complexity of Payload Conjugation: The incorporation of two cytotoxic agents with distinct mechanisms of action can introduce complex metabolic pathways and elevate the risk of toxicity. Maintaining antibody stability, target specificity, and controlled payload release becomes significantly more challenging in dual-payload ADC design.
- 5.2 Optimizing Drug-to-Antibody Ratio (DAR): Determining the optimal number and balance of payloads required for effective tumor cell killing—while preserving manufacturability and patient tolerability—remains a major hurdle. Elevated DARs can increase adverse effects, and structural antibody modifications may compromise stability and increase immunogenicity. Additionally, achieving consistent conjugation ratios and product uniformity presents significant manufacturing challenges.
- 5.3 Managing Toxicity and Off-Target Effects: The administration of dual cytotoxic payloads increases the risk of off-target activity, cytokine release, and systemic toxicity. Achieving tumor-restricted delivery while minimizing exposure to healthy tissues is therefore critical for clinical success.
- 5.4 Manufacturing and Regulatory Barriers: Dual-payload ADCs represent a relatively new and complex therapeutic class, requiring extensive analytical characterization, stability testing, and tighter risk margins during early clinical development. Compared to single-payload ADCs, production involves additional reaction steps, orthogonal conjugation handles, and post-protein synthesis modifications, all of which add substantial complexity to manufacturing and regulatory approval processes.
6. Future Directions in ADC Therapy
Multi-payload antibody–drug conjugates (ADCs) are emerging as a promising evolution in targeted cancer therapy, offering the potential for greater therapeutic efficacy, reduced drug resistance, and improved safety profiles. By combining two cytotoxic agents with distinct mechanisms of action, dual-payload ADCs can overcome resistance pathways, address tumor heterogeneity, and deliver more durable clinical responses compared to traditional single-payload constructs.
Ongoing advances in site-specific conjugation techniques, orthogonal linker systems, and non-canonical amino acid incorporation are enabling higher precision, improved linker stability, and enhanced scalability in ADC manufacturing. These technological innovations are critical for translating dual-payload ADCs into robust and reproducible clinical products.
Beyond oncology, dual-payload ADCs are expected to unlock new therapeutic opportunities in autoimmune disorders and infectious diseases, where the selective delivery of multiple therapeutic agents could fundamentally transform treatment paradigms. Supported by growing industry investment and early-stage clinical programs, dual-payload ADCs are well positioned to expand into new application areas, delivering meaningful scientific innovation with real-world therapeutic impact.
7. Conclusion
Dual-payload ADCs provide a multidimensional strategy for cancer treatment by simultaneously addressing tumor heterogeneity, therapeutic resistance, and safety concerns. Their success relies on precise linker chemistry, optimized drug-to-antibody ratios, and controlled payload release, with emerging innovations such as bifunctional and PEGylated linkers already demonstrating improved performance.
Although challenges related to manufacturing complexity, regulatory requirements, and long-term safety remain, continued innovation positions dual-payload ADCs as a highly promising platform capable of redefining targeted cancer therapy through enhanced efficacy and superior control over drug delivery.



-Sensors-Next-Generation-Flexible-Devices/Wrinkled-Silver-Nanowire-AgNW-Sensors-Next-Generation-Flexible-Devices.png)






-Innovative-Applications-and-Insights.jpeg)