1. Introduction
Plasma medicine is a rapidly growing field that leverages the principles of plasma science and engineering for various biomedical applications. Plasma, often referred to as the fourth state of matter, is formed when energy, such as heat, causes gas atoms to lose electrons by ionization, resulting in an ionized gas that conducts electricity and responds to magnetic fields.
Two major plasma treatment modalities exist:
Plasma-activated liquids (PALs) are created by exposing liquids—such as deionized water, saline, or culture media—to cold atmospheric plasma (CAP). This process generates a complex mixture of reactive oxygen and nitrogen species (RONS) within the liquid, which are the primary mediators of PALs’ biological activity.
Chemically, PALs are generated through a three-phase transfer of reactive species—from gas → interface → liquid. The interaction of plasma with the surrounding gas and liquid phase initiates a cascade of reactions, ultimately forming a variety of short- and long-lived RONS. Among these, the key long-lived species include:
- Reactive Oxygen Species (ROS): Hydrogen peroxide (H₂O₂), singlet oxygen (¹O₂), ozone (O₃)
- Reactive Nitrogen Species (RNS): Nitrite (NO₂⁻), nitrate (NO₃⁻), nitric oxide (NO)
These reactive species are fundamental to the antimicrobial, anti-inflammatory, and anticancer effects observed with PAL treatment. Importantly, the composition and concentration of RONS within PALs can be precisely tuned by adjusting plasma parameters such as gas composition, applied voltage, exposure duration, and the distance between the plasma source and liquid surface.
Owing to their stability, ease of storage, and versatility, PALs are gaining increasing attention in diverse domains including infection control, wound healing, oncology, agriculture, food safety, and environmental decontamination. In medical applications, they are particularly valuable in scenarios where direct plasma exposure is not feasible, enabling targeted delivery of reactive species in hard-to-reach anatomical sites.
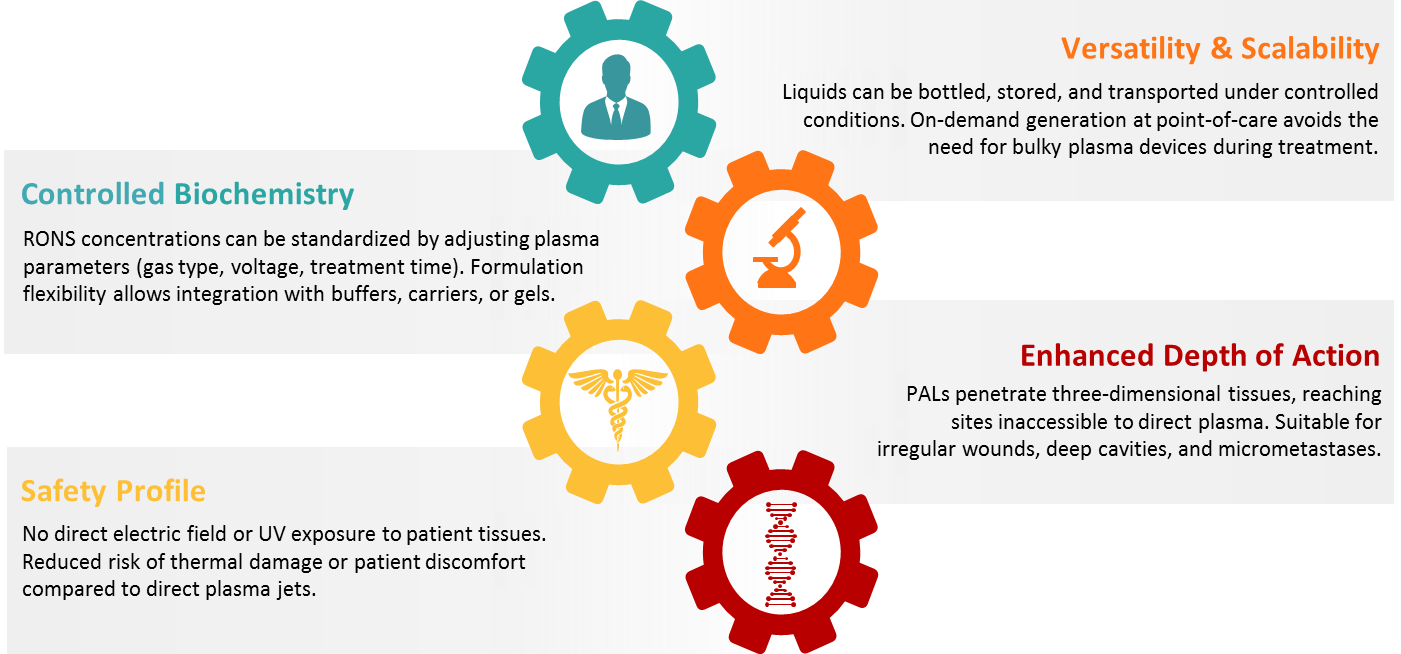
2. Benefits of PALs
3. Advantages of Indirect Plasma Treatment
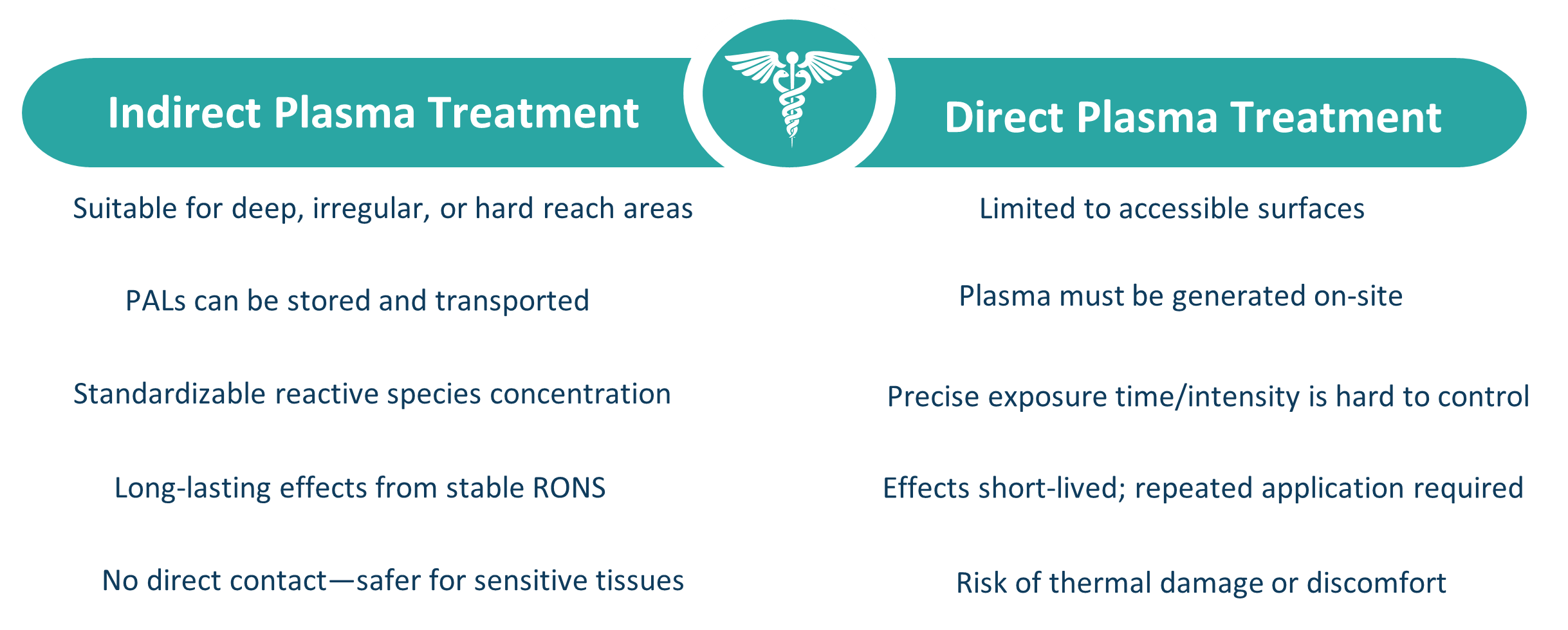
4. Limitations of Direct Plasma Treatment
The use of plasma in medical applications is currently limited due to several factors associated with its direct application.
- Plasma treatment typically affects only the outermost surface layer (approx. 10 nm). However, penetration into deeper tissues such as muscles, bones, or organs is limited and not yet fully understood. While some studies suggest that reactive species can penetrate the stratum corneum (the skin’s main barrier), and others have shown anticancer effects in tumors implanted under the skin of mice, the depth of penetration remains inadequate, and the underlying mechanisms are not fully understood.
- Direct plasma application is generated by a device, meaning the device must be physically present at the location where the plasma is needed. This limits plasma's use based on the availability and configuration of the device. Furthermore, it is difficult to sustain or modify the plasma once it is generated, as it is produced directly by the device.
In contrast, PALs enable more consistent and standardized dosing by maintaining controlled concentrations of reactive species, pH, and redox potential, overcoming many of these direct-application challenges.
5. Methods for Generating PALs
Various systems are available for generating plasma-activated liquids, each characterized by different properties. However, the way plasma interacts with the liquid is especially crucial for PALs, as it significantly impacts the resulting plasma characteristics.

5.1 Plasma over Liquid Surface (Indirect Contact)
One of the most common methods used for producing plasma-activated liquid includes a plasma discharge generated in the gas phase which then comes into contact with the water surface. Two commonly used systems for generating PALs include dielectric barrier discharge (DBD) devices and plasma jets, both the systems have distinct advantages. In DBD systems, plasma ignites in a gap between a high-voltage electrode and the target material, which results in the direct contact of the plasma and the target. Alternatively, plasma can be generated on the surface of a specially designed electrode structure that is isolated from a counter electrode. However, in plasma jets, plasma is created inside a capillary or tube within a pen-like device, which houses a specific electrode setup for plasma generation.
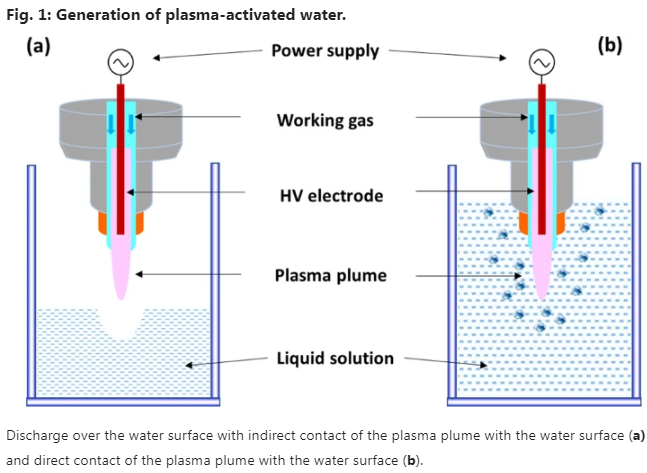
Non-touch conditions mean that cold atmospheric plasmas (CAPs) are generated at a distance from the water surface, meaning there is no direct contact between the visible gas-phase plasma and the aqueous solution. Under these conditions, reactive oxygen and nitrogen species (RONS) are first generated in the gas phase, and only the more stable, long-lived species are able to dissolve into the solution. However, in the direct contact mode, the CAPs are generated within the water. In addition to the reactive species that accumulate from the gas-phase plasma, a range of other reactive species is formed through plasma-liquid interactions within the solution itself.
5.2 Bubble or Multiphase Plasma Systems
Multiphase plasmas are used to increase the chemical reaction rates of plasma-induced species interacting with liquids and to improve the transfer of these species from the gas phase to the liquid phase, thereby enhancing the performance of existing plasma-based systems. Multiphase plasmas are generated within bubbles or ignited in the gas phase, which is then mixed with water droplets.
For discharge within bubbles, a surface streamer is triggered inside a forming gas bubble within a liquid. The discharge travels along the gas-liquid interface, generating significant amounts of reactive oxygen and nitrogen species (RONS), which in turn enhances mass transfer rates. These bubbles function as carriers, transporting RONS from the plasma into the liquid, enhancing PALs activity.
5.3 Direct Discharges in Liquid (High-Field Systems)
Direct liquid phase discharge is a process in which the plasma is generated directly within the bulk solution. In this setup, a high-voltage electrode, typically in the shape of a needle or plate, is submerged in the liquid. Compared to discharges in gases, those in water require a much stronger electric field (around 1 MV/cm) due to the increased density of water molecules, which makes it harder for electrons to move through water than through air. As a result, the formation of bubbles in the liquid will absorb some of the energy, reducing the overall input compared to a discharge directly in the water. The key characteristics of direct liquid phase discharges include a high electron density, elevated gas temperatures, and high discharge currents.
It is important to note that, in addition to the plasma-generating systems, the characteristics of the plasma are also influenced by several other factors related to the plasma source. These include input power, treatment voltage, the treatment mode, and the gap between the plasma plume and the liquid surface.
6. Applications of PALs
The unique properties of PALs make them suitable for a range of applications across healthcare, agriculture, and environmental fields.
6.1 Sterilization and Disinfection
Plasma-activated liquids (PALs) have demonstrated broad-spectrum antimicrobial activity, effectively inactivating bacteria, fungi, and viruses. This makes PALs a promising tool for sterilization and disinfection, particularly in medical, industrial, and environmental settings.
The mechanism of action of PALs against bacteria differs based on cell wall structure:
- In Gram-negative bacteria, reactive oxygen and nitrogen species (RONS) induce lipid peroxidation in the outer membrane. This leads to the formation of malondialdehyde (MDA), a byproduct known to interact with and damage DNA, ultimately contributing to cell death.
- In contrast, Gram-positive bacteria are more susceptible to direct intracellular damage caused by RONS, rather than through lipid peroxidation, due to the thicker peptidoglycan layer in their cell walls.
Biofilm inactivation is a major focus of recent research. Biofilms present a significant challenge in healthcare and industry due to their resistance to conventional antibiotics and disinfectants. They are commonly implicated in hospital-acquired infections—such as on catheters, prosthetic devices, and ventilators—and are notoriously difficult to eradicate.
One key advantage of PALs over gas-phase plasma is its ability to treat biofilms without causing drying. Plasma-activated water (PAW) retains moisture while delivering reactive species, thereby maintaining biofilm permeability and increasing susceptibility to antimicrobial action. The complexity of chemical interactions between RONS in PALs and components of the biofilm matrix is beneficial—producing a diverse mix of short- and long-lived species that collectively disrupt microbial viability.
These findings support the use of PALs in a variety of sterilization scenarios, including the disinfection of medical instruments, surfaces, and even human tissue, where minimizing collateral damage and moisture loss is critical.
6.2 Anticancer Applications
Emerging evidence suggests that plasma-activated liquids (PALs) possess strong anti-cancer properties. Both in vitro and in vivo studies indicate that PALs can induce selective cytotoxicity in cancer cells, primarily through apoptosis, without significantly harming normal, healthy cells.
- In a xenograft tumor model involving paclitaxel/cisplatin-resistant ovarian cancer cells, subcutaneous injections of plasma-treated RPMI-1640 medium led to a significant reduction in tumor volume and weight over a 28-day period. These results suggest that PALs may offer a complementary or alternative strategy for drug-resistant tumors.
- Another study demonstrated the ability of PALs to selectively trigger apoptosis in cancer cells, sparing normal cells when properly optimized. A portable plasma pen using a streamer corona discharge in ambient air was employed to activate phosphate-buffered saline (PBS) and cell culture medium. The concentrations of hydrogen peroxide (H₂O₂), nitrite (NO₂⁻), and nitrate (NO₃⁻) were measured, revealing a nearly 1:1 ratio of H₂O₂ to NO₂⁻—a balance considered crucial for initiating cancer cell apoptosis.
Notably, this balanced reactive species profile is a unique feature of PALs produced via air-based plasma pens, in contrast to those generated using traditional argon plasma jets. This makes air-based systems more accessible, cost-effective, and potentially scalable for therapeutic use.
These studies highlight the potential of PALs as a non-invasive, tunable anticancer strategy—especially valuable in addressing chemotherapy resistance and reducing systemic toxicity.
6.3 Tissue Regeneration
Plasma-activated liquids (PALs) show promising potential in enhancing wound healing and deep tissue regeneration. Due to their liquid nature, PALs can be easily applied to irregular wound surfaces, providing practical advantages over conventional solid treatments.
- In muscle tissue regeneration, a study demonstrated that exposure of myoblast cells to culture medium treated with nitrogen–argon (N₂Ar) plasma led to increased expression of muscle differentiation markers, including myosin heavy chain (MyHC) and myogenin. This suggests its possible application in muscle repair therapies.
- In the context of the central nervous system, PALs have shown regenerative effects on astrocytes, which are supportive glial cells involved in neuroinflammation and repair. Culture media treated with N₂, air, or O₂ plasma were applied to astrocytes in a scratch wound healing assay. All three types of PALs significantly enhanced both cell migration and proliferation, indicating that PALs can promote neural tissue regeneration.
An emerging and innovative strategy involves the use of plasma-treated hydrogels. Hydrogels are three-dimensional, semi-solid networks composed of hydrophilic polymers that can hold substantial amounts of water, making them ideal carriers for PAL. Plasma treatment not only aids in the formation and cross-linking of hydrogel structures but also allows these materials to generate, retain, and gradually release reactive oxygen and nitrogen species (RONS).
In vitro studies have demonstrated that such plasma-treated hydrogels possess sustained antibacterial activity, making them suitable for use as advanced wound dressings or tissue scaffolds in regenerative medicine.
7. Conclusion and Future Outlook
Plasma continues to play a crucial role in driving innovation in modern medicine. Among the various methods, indirect plasma treatment, particularly through the use of plasma-activated liquids (PALs), stands out for its numerous advantages over direct plasma application. Indirect methods are non-invasive and safer for sensitive tissues, as they avoid direct contact with the plasma discharge, thereby minimizing the risks of thermal damage and patient discomfort.
The biological activity of PALs is largely attributed to the presence of reactive oxygen and nitrogen species (RONS). Depending on the plasma source, treatment parameters, and liquid composition, both short-lived RONS (such as hydroxyl radicals and singlet oxygen) and long-lived RONS (such as hydrogen peroxide, nitrites, and nitrates) can be generated. Short-lived species are highly reactive and act immediately upon application, while long-lived species allow for sustained biological effects, enabling PALs to be stored and transported for later use. This adaptable reactivity offers a versatile platform for a wide range of biomedical applications—from antimicrobial treatments and biofilm disruption to cancer therapy and tissue regeneration.
Future Research Directions
Plasma-activated liquids (PALs) show immense potential, but several challenges and opportunities remain:
- Mechanistic Clarity: While PALs generate reactive oxygen and nitrogen species (RONS) that can induce cell death or modulate immune responses, the exact molecular pathways—especially differences between short- and long-lived RONS—are still not fully understood. Further research is needed to clarify these mechanisms.
- Clinical Translation: Preclinical models have shown promising results in cancer, infection, and wound healing. Small-scale human trials show safety, but larger, controlled studies are essential to validate efficacy and guide regulatory approvals.
- Formulation and Stability: PALs degrade quickly due to the reactive nature of RONS. Solutions include on-demand generation, buffer stabilization, or integrating PALs into gels or delivery systems. Portable plasma generators and standardized protocols are needed for clinical use.
By focusing on these areas, future work can move PALs from experimental platforms to practical, safe, and effective biomedical tools.