Biofabricated Skin: Next-Generation Wound Healing and Cosmetic Applications
Executive Summary
The transformative potential of biofabrication as a next-generation solution for wound healing, burn treatment, and cosmetic dermatology. Traditional skin grafts face limitations such as scarring, donor site morbidity, and immune rejection, while biofabricated skin offers functional, aesthetic, and regenerative advantages by incorporating living cells, biomaterials and advanced fabrication techniques like 3D bioprinting, electrospinning, and organoid engineering. Key innovation include vascularized constructs, smart drug-delivery systems, and personalized pigmented grafts. Applications span from chronic wound care to scar revision and in vitro cosmetic testing. Future directions emphasize achieving full biomimicry, neural integration, scalability and global accessibility positioning biofabricated skin as a cornerstone in regenerative medicine and aesthetic reconstruction.
1. Introduction
The human skin is the complex, outermost and multi-layered covering of the body and is the largest organ of the integumentary system. The skin has up to seven layers of ectodermal tissue guarding muscles, bones, ligaments and other internal organs. Skin is a complex, multilayered organ comprising Epidermis, dermis, hypodermis with multiple cell types such as collagen, melanocytes, keratinocytes, fibroblasts, elastin, nerves and sweat glands. Skin plays an important role in protection, thermoregulation, and synthesis of vitamin D and regulation of vitamin B.
Skin injuries, ranging from acute burns to chronic wounds, affect millions worldwide and pose significant challenges to both healthcare systems and patients. Traditional skin grafts generally leads to scarring due to the lack of hair follicles and sweat glands and donor sites can suffer pain or additional scarring especially in large injuries. In case of severe burns, limited donor skin complicates the treatment, while allografts pose a risk of immune rejection and infection. Biofabricated skin offers a promising alternative for replicating the structure and function of the skin using live cell, scaffolds and bioactive molecules. Techniques like 3-D printing and stem cell engineering enables the creation of multi-layered, vascularized skin construct. These innovation support natural healing, minimize scarring and improved outcomes. As such, biofabricated skin is revolutionizing care in wound healing, burns, reconstruction, and cosmetic dermatology.
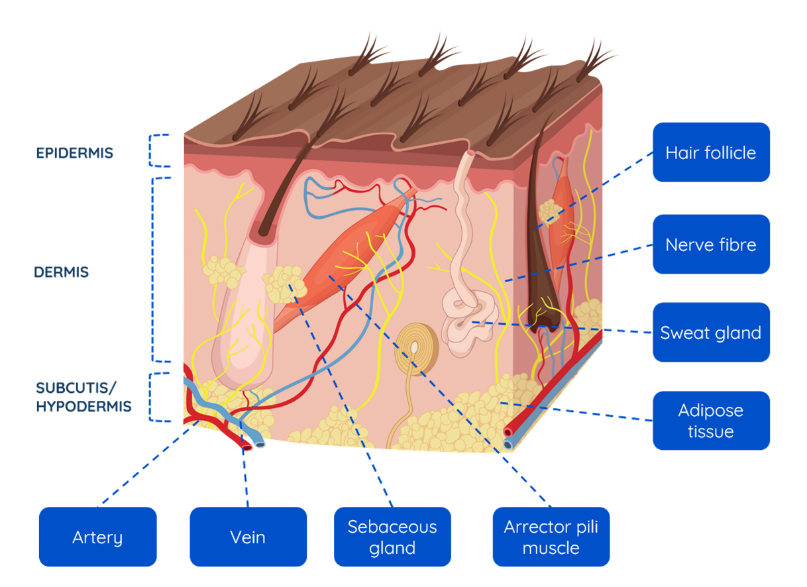
Figure 1. Different layers of skin
2. Skin Biofabrication
Skin biofabrication is the process of creating artificial skin using live cells and biomaterials to replicate natural skin structure and function. It involves technologies like 3D bioprinting, where layer of cells and scaffolds are precisely deposited to form skin tissue. This engineered skin can be used for treating burns, wounds and skin diseases, offering an alternative to traditional grafts. The goal is to produce skin that integrates well with the body and promotes healing.
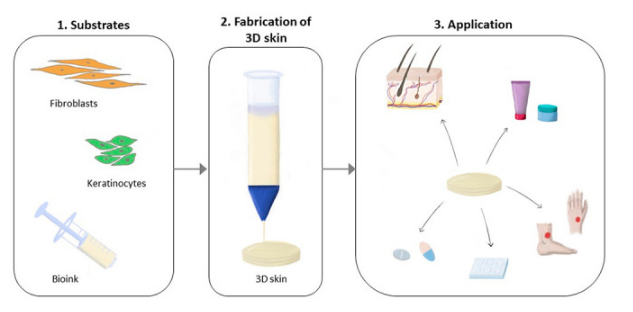
Figure 2. Process of Biofabrication
3. Problem: Limitations of Traditional Skin Repair
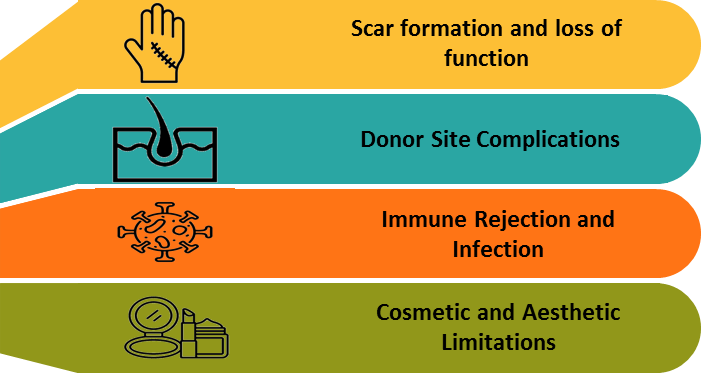
3.1. Scar Formation and Loss of Function
Traditional skin grafts are unable to restore key appendages such as sweat gland, hair follicles and melanocytes. As a result, the healed skin often forms rigid, discolored scars that lack the functionality of natural skin. This leads to impaired thermoregulation and reduced sensory capabilities with poor outcomes which can lead to the decreased quality of life.
3.2. Donor Site Complications
Autografting involves removing skin from healthy areas of the patient's body, which can create additional wounds and complications. This process adds to patient discomfort and prolongs recovery time. In case of extensive burns, the limited availability of donor skin poses a major challenge. The shortage can delay critical treatment and heighten the risk of infection.
3.3. Immune Rejection and Infection
Allografts and Xenografts often provoke immune responses in the recipients, demands the use of immunosuppressive therapies. Despite these measures, the body may still reject the graft over time. These types of grafts are generally used as temporary solution rather than permanent fixes. Their susceptibility to infection and rejection further limits their long-term effectiveness.
3.4. Cosmetic and Aesthetic Limitations
Despite successful wound closure, traditional skin repair methods often fall short in delivering satisfactory aesthetic outcomes. Scarring is common, along with mismatched textured and an absence of natural pigmentation. These visible differences can be especially troubling for patients, particularly when affecting prominent areas. The resulting appearance may lead to a loss of confidence and self-consciousness. Over time, such issues can contribute to significant psychosocial stress and decreased quality of life.
4. The Solution: Biofabricated Skin
Biofabricated skin is an advanced, lab-engineered construct designed to closely imitate the structure and function of natural human skin. It is developed using a combination of living cells such as keratinocytes, fibroblasts, melanocytes, endothelial cells, and stem cells which work together to replicate the behavior of real skin. These cells are supported by biometric scaffolds made from natural or synthetic extracellular matrix (ECM) materials that provide structural integrity and guide tissue regeneration. Biofabricated skin also incorporates essential growth factors and bioactive molecule to enhance healing and tissue regeneration. Unlike traditional grafts, it is capable of including functional elements like sweat glands, hair follicles and nerves. This allows it to restore both the appearance and full functionality of damaged skin. As a result, biofabricated skin represents a major leap forward in regenerative medicine and wound care.
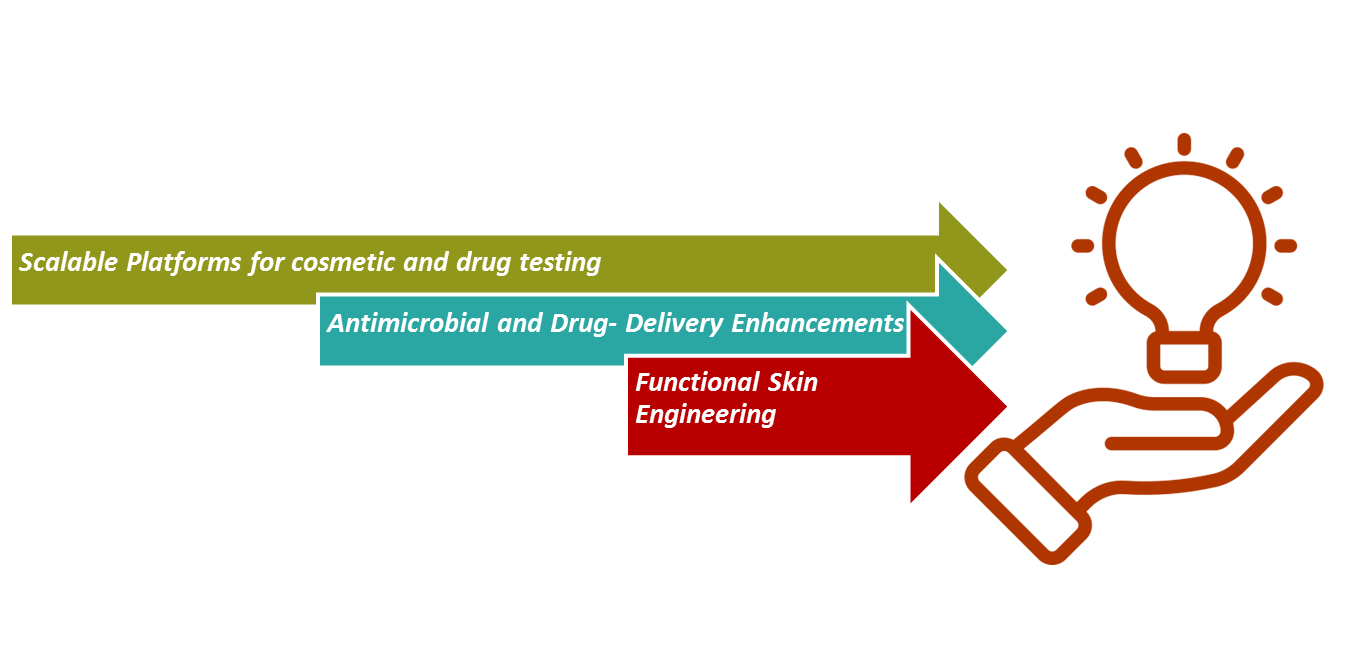
4.1. Functional Skin Engineering
Sweat glands and hair follicles are successfully integrated using organoid culture and bio-ink innovation. Pigmentation can be achieved through the inclusion of melanocytes during fabrication, enabling aesthetic and UV-protective features.
4.2. Antimicrobial and Drug-Delivery Enhancements
Embedding antimicrobial peptides directly into the skin matrix to prevent infection. Smart skin grafts are capable of controlled drug release such as antibiotics or anti-inflammatory agents. Use of genetically engineered skin cells that respond to environmental indication for targeted therapy.
4.3. Scalable Platforms for Cosmetic and Drug Testing
Development of standardized, reproducible skin models for toxicity and efficacy testing. Replacement of animal models with physiologically relevant human skin. Ability to mimic specific condition (e.g., acne, eczema, pigmentation disorders) for tailored testing.
5. How Biofabricated Skin Overcomes Traditional Challenges
| Challenge | Traditional Grafting Limitation | Biofabricated Skin Solution |
|---|---|---|
| Unlimited Supply via In Vitro Production | Limited donor skin availability, especially in large burns and severe wounds. | Skin is grown from a small biopsy of the patient's own cells, offering an unlimited, lab-generated source and eliminating donor site complications. |
| Reduced Immune Rejection | Allografts and Xenografts are often rejected by the immune system, requiring immunosuppressive drugs. | Autologous biofabricated skin (from the patient's own cells) minimizes immune rejection, reducing or eliminating the need for immunosuppression. |
| Enhanced Healing and Integration | Grafts may not adhere well or vascularize efficiently, leading to poor healing and graft failure. | Engineered with growth factors, stem cells, and vascular structures to support faster healing, better integration and long-term viability. |
| Customization and Precision | Traditional grafts cannot be tailored for pigmentation, thickness, or structure. | Bioprinting allows layer-by-layer control, enabling personalized skin substitutes that match the patient's tone, thickness, and potentially includes appendages. |
| Cost and Scalability | Limited scalability, high surgical burden, and resource-intensive donor harvesting. | Automation and advances in bioink technologies are making large-scale production feasible, improving cost-efficiency over time. |
6. Different Biofabrication Techniques
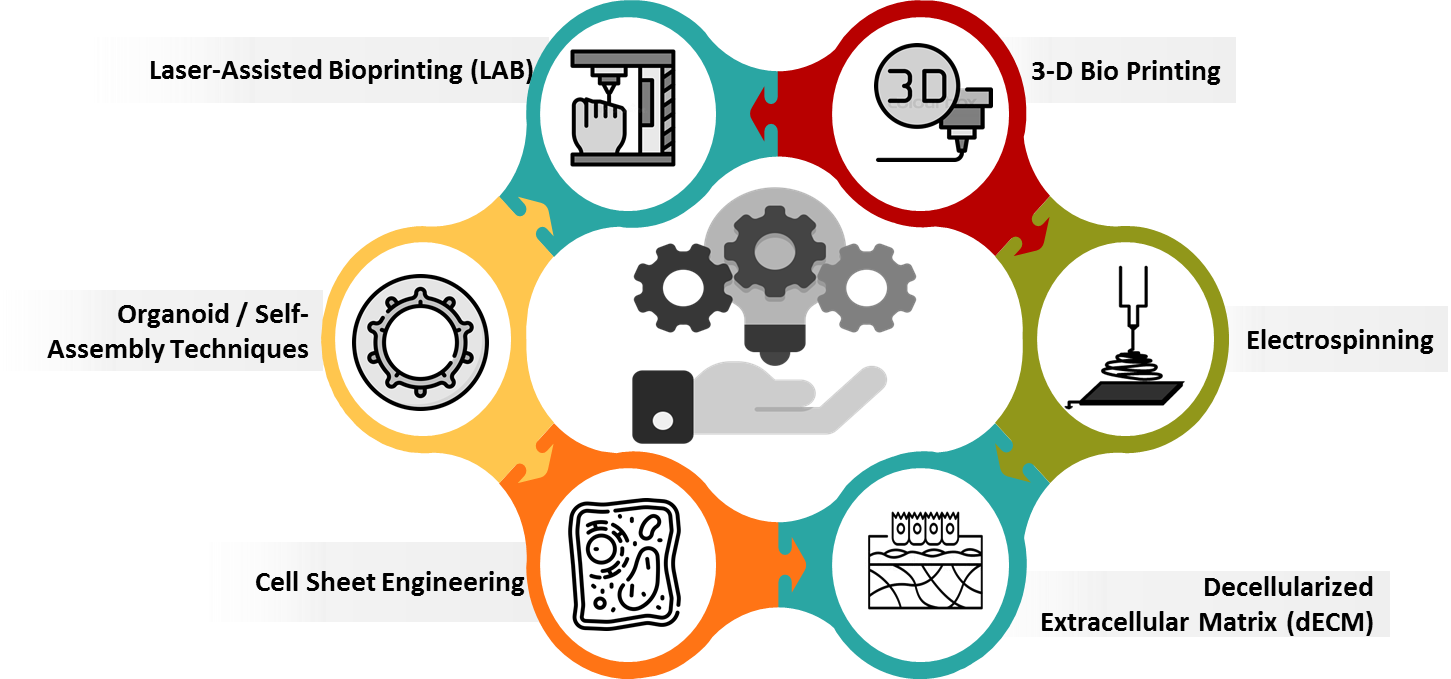
6.1. 3-D Bio Printing
It refers a manufacturing process where bioinks composed of cells, hydrogels and biomaterials are specifically deposited layer by layer to replicate the structure of native skin. It allows high control over spatial organization, enabling the fabrication of multi-layered skin with separate epidermal and dermal components. Techniques include extrusion-based (good for thick, cell-filled constructs), inkjet (high-resolution but limited viscosity), and laser-assisted (precise droplet placement with nozzles). This method enables the creation of multi-layered skin, replicating dermis, epidermis and even vasculature or hair-follicles. Bio printing supports customization for patient-specific applications and holds promise for grafting large wounds. However, challenges remain in maintaining cell viability, achieving functional integration and ensuring vascularization post-implantation.
6.2. Electrospinning
Electrospinning uses a high-voltage electric field to draw ultrafine polymer fibers from a liquid solution, forming nanofibrous mats that resemble the extracellular matrix (ECM) of native skin. These mats provide an ideal scaffold for cell attachment, proliferation, and migration, especially for dermal reconstruction. Electrospun scaffolds can be fabricated using synthetic polymers (like PCL, PLA) or natural ones (like collagen, silk fibroin), often combined to balance mechanical strength and bioactivity. They can also be functionalized with antimicrobial agents or growth factors for improved healing.
6.3. Decellularized Extracellular Matrix (dECM)
This technique involves removing cells from donor skin (human or animal) through chemical, enzymatic or physical processes, leaving behind a biochemically rich ECM scaffold. dECM maintains native structural proteins, cytokines, and growth factors essential for cell signaling, adhesion, and tissue regeneration. These scaffolds can be used directly or processed into hydrogel bioinks for bio printing. Recellularization with patient-derived cells minimizes immune responses and enhances graft integration. Decellularized extracellular matrix exhibits excellent biocompatibility.
6.4. Cell Sheet Engineering
In this scaffold-free method, cells are cultured into continuous sheets that preserve their native extracellular matrix and cell-cell junctions. These sheets can be layered to reconstruct epidermis without using synthetic scaffolds. The method uses temperature-responsive surfaces to harvest sheets without damaging the extracellular matrix. Cell sheet integrate well with host tissue and show high viability and function, making them useful for burns, ulcers, and corneal repair. Combining cell sheet with support materials can overcome their limitation of manipulation, improving structural integrity for deep wounds.
6.5. Organoid / Self-Assembly Techniques
Such as iPSCs (induced pluripotent stem cells) or ESCs (embryonic stem cells) or progenitor cells to self-organize into 3D skin-like structures under guided conditions. Skin organoids can replicate key physiological components like the epidermis, dermis, melanocytes, and sometimes even hair follicles and sweat glands. Organoids offer a biologically accurate model for disease studies, drug testing, and future personalized grafts. They are often limits by long culture times, batch variability, and difficulties in scaling for large wound coverage.
6.6. Laser-Assisted Bioprinting (LAB)
Laser assisted bioprinting method that uses a pulsed laser to push cell scaffold bioink droplets from a donor ribbon onto a collector surface. This allows precise placement of single cell or cell clusters without the nozzle-induced shear stress seen in other methods. It's suitable for building multi-layered skin structures with accurate cell arrangement and high-viability. The lack of direct contact reduces contamination risk and enhances cell survival, but the setup is costly, complex, and limited in throughput making it more suitable for research and small-scale fabrication than clinical use at present.
7. Key Innovation and Recent Advancements

7.1. Vascularized 3D-Printed Skin Constructs
A Swedish team developed artificial skin using two complementary 3D bioprinting approaches: one with a bioink composed of fibroblasts embedded in gelatin grains in a hyaluronic acid gel, and another method called refresh, which uses hydrogel filaments that form customizable, enzyme-removable channels acting as a blood vessels. In vivo mouse studies showed dermis regeneration and vascularization in implanted grafts. It is considered as a major advancement because vascularization is a long-standing barrier in thick skin constructs.
7.2. Hybrid Bioinks with Adipose Derived ECM and Alginate
A recent study (2025) introduced a "hybrid bioink" made of adipose-derived decellularized extracellular matrix and alginate. It was optimized to support adipogenesis, proper nutrient/oxygen diffusion, reduce unwanted migration of precursors, and when assembled with dermis modules, promoted wound healing in mice via re-epithelization, tissue remodeling, and angiogenesis. This shows promise in integrating adipose tissue biofabrication into skin grafts.
7.3. Photo-Activated Silk-Collagen-like Protein Hydrogel ("PASCH")
Researchers in India developed a lab-made collagen like protein hydrogel, activated by visible blur light and riboflavin to form stable crosslinks without harmful UV or toxic chemicals. The gel called PASCH, is porous, flexible, retains moisture, helps reduce inflammation, and accelerates wound closure. It's being modified especially for diabetic wounds and burns.
7.4. Three-Layer ECM-Based Biogel Skin Substitute
A collaboration in Spain engineered a 3-layer biogel based on extracellular matrix components to mimic epidermis, dermis and hypodermis. It serves as a bioactive skin substitute that accelerate regeneration, offering better architecture and skin layer modeling than simpler scaffolds.
7.5. Self-Healing Hydrogels Inspired by Skin Mechanics
Researchers from Aalto and Bayreuth developed hydrogels that combine strength, flexibility, and autonomous self-healing. By integrating ultra-thin clay nanosheets with entangled polymer networks, they achieved materials that recover from damage, mimic some mechanical properties of human skin, and may be used in dressings, artificial skin, or in responsive wound healing applications.
8. Key Applications
| Application | Specifications |
|---|---|
| Treatment of Chronic Wounds |
|
| Burn Injury Management |
|
| Temporary and Permanent Skin Grafts |
|
| Scar Reduction and Functional Restoration |
|
| Vascularized Skin Substitutes |
|
| Anti-Aging Skin Therapies |
|
| Scar Revision and Skin Resurfacing |
|
| Regenerative Skin Patches |
|
| 3D Bio-printed Cosmetic Skin Models |
|
| Pigmented Skin Grafts |
|
9. Future Directions
Biofabricated skin in wound healing and cosmetics aims to enhance its functionality, accessibility, and clinical translation. A major focus is on achieving full biomimicry by incorporating skin appendages like hair follicles, sweat glands, melanocytes, and nerves to restore both form and function. Advanced research is exploring immune-responsive grafts and neural integration for better physiological response and sensory restoration. Vascularization remains a critical challenge, driving the development of pre-vascularized constructs and rapid host integration strategies, supported by scalable 3D Bioprinting and automation. Smart skin constructs with self-healing hydrogels, embedded biosensors, and stimuli-responsive drug delivery systems are also being developed to improve healing and reduce complications. Cosmetically, there is a push for pigmented, scar-free grafts customized for aesthetic and reconstructive needs, such as vitiligo, facial burns and pigmentation disorders. Integrating adipose tissue and building multi-layered constructs with volume restoration capabilities enhances realism and function. Ethical and regulatory progress is needed through GMP-compliant manufacturing, standardized in vitro skin models, and long-term clinical trials. Personalized solutions are emerging via skin organoids and induced pluripotent stem cell derived grafts for disease modeling and tailored treatment. Hybrid biofabrication strategies combining bioprinting, electrospinning, dECM, and cell sheets are optimizing graft structure. Lastly, innovations are focusing on global accessibility through low-cost skin kits and portable bioprinters for use in case of low-resource and emergency. These direction aim to redefine skin regeneration and cosmetic dermatology by merging regenerative medicine, bioengineering and personalized care.
10. Conclusion
Biofabricated skin represents a groundbreaking advancement in regenerative medicine, offering a viable and superior alternative to traditional grafting techniques. By combining live cells, bioactive scaffolds and advanced fabrication technologies, it enables the creation of skin constructs that restore both the appearance and full functionality of damaged tissue. These innovations address key limitation of current treatments including scarring, donor site complications, and immune rejection while opening new possibilities in wound care, aesthetics and drug testing. As research advances towards fully biomimetic, vascularized, and sensory-integrated skin, the clinical and cosmetic potential of biofabricated skin continues to expand. With ongoing efforts in personalization, scalability, and accessibility, biofabricated skin is redefining the future of skin repair and dermatological therapy.



-Sensors-Next-Generation-Flexible-Devices/Wrinkled-Silver-Nanowire-AgNW-Sensors-Next-Generation-Flexible-Devices.png)

