Novel Purification Techniques for Human IgG
Executive Summary
In this Paper we will discuss about the background of the novel purification Techniques for Human IgG. In which old and traditional method will be discussed how they are costly Techniques for Purification of Human IgG and after that they do not provide 95 to 97% purity and yield of human IgG. They are very time consuming method and having so much process is doing for one step. And further we will discuss about novel and modern techniques of the purification of human IgG method. And also discuss about the comparison and selection between the best methods for purification and challenges and limitations of the purification methods of human IgG.
1. Introduction
Human Immunoglobulin G is important component of the adaptive immune system and show one of the most clinically significant and commercially valuable class of therapeutic proteins and widely used as therapeutically for treating immunodeficiency, autoimmune diseases and infections. IgG plays an important role in immune defense mechanisms, including neutralization of pathogens, activation of complement pathways and mediation of antibody-dependent cellular cytotoxicity.
Purification is very important and critical step in ensuring the safety and efficacy of IgG therapies. In traditional purification method such as Cohn cold ethanol fractionation method, ion exchange chromatography and protein A affinity chromatography they show some numerous challenges when scaled at industrial production. They have limitations regarding to high operational costs, extended the processing times, relatively low yields and environmental concerns because due to the high use of organic solvents and generation of hazardous waste. These conventional methods may struggle to achieve higher purity levels of beyond 97% and it comes to removing closely related impurities such as IgA, IgM, and human serum albumin.
This paper focuses on identifying and evaluates the advanced purification strategies to enhance the industrial-scale processing of human IgG and aim for improving the product quality, reduced environmental impact and greater operational efficiency.
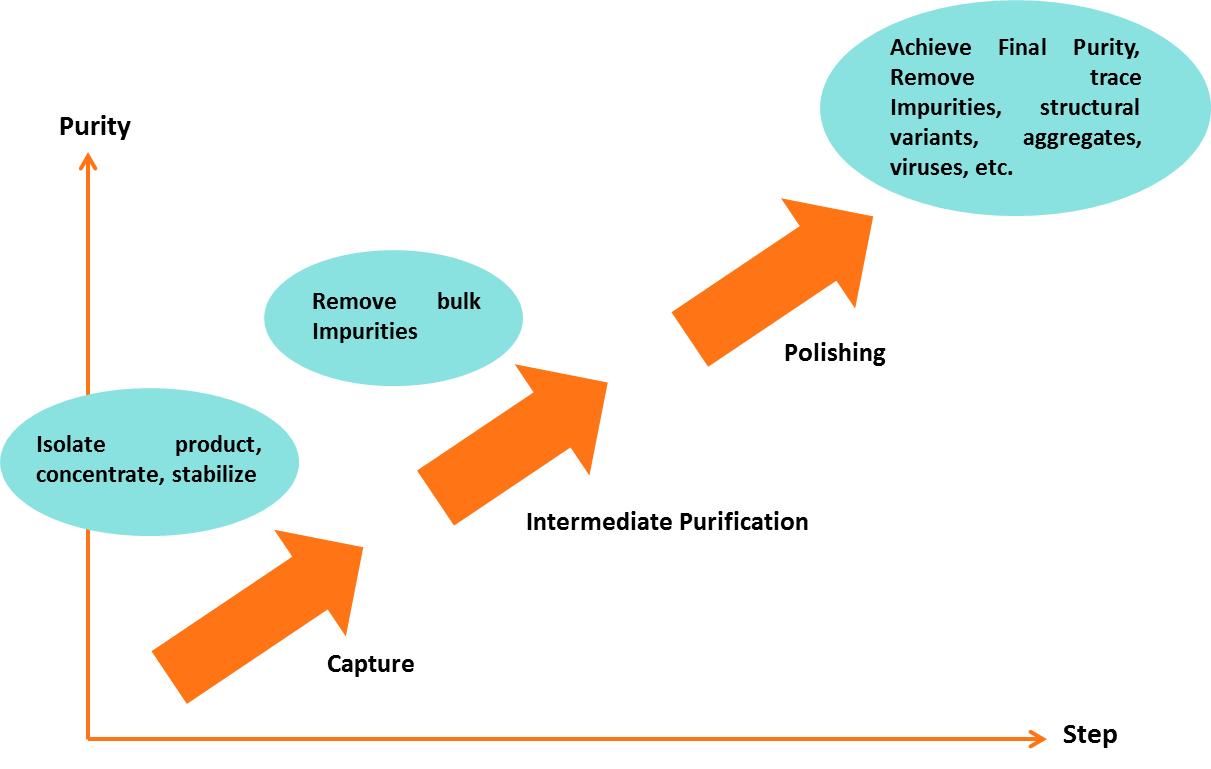
2. Three Phase Strategy for Purifying Antibodies
3. Conventional VS Modern Purification Methods
Cohn's fractionation (cold ethanol fractionation) is traditional and old technique to purify or separate IgG from Human Plasma. It is done in five fractions steps mainly. After separation of plasma proteins, Fraction II step contains IgG. This technique step shows that high % of yielding loss of IgG. Old Conditions of this technique are following (Figure 1). In modern technique use of chromatographic for purification of fraction II have avoid this loss. In this modern purification technique improved elution is observed and shown in (Figure 2).
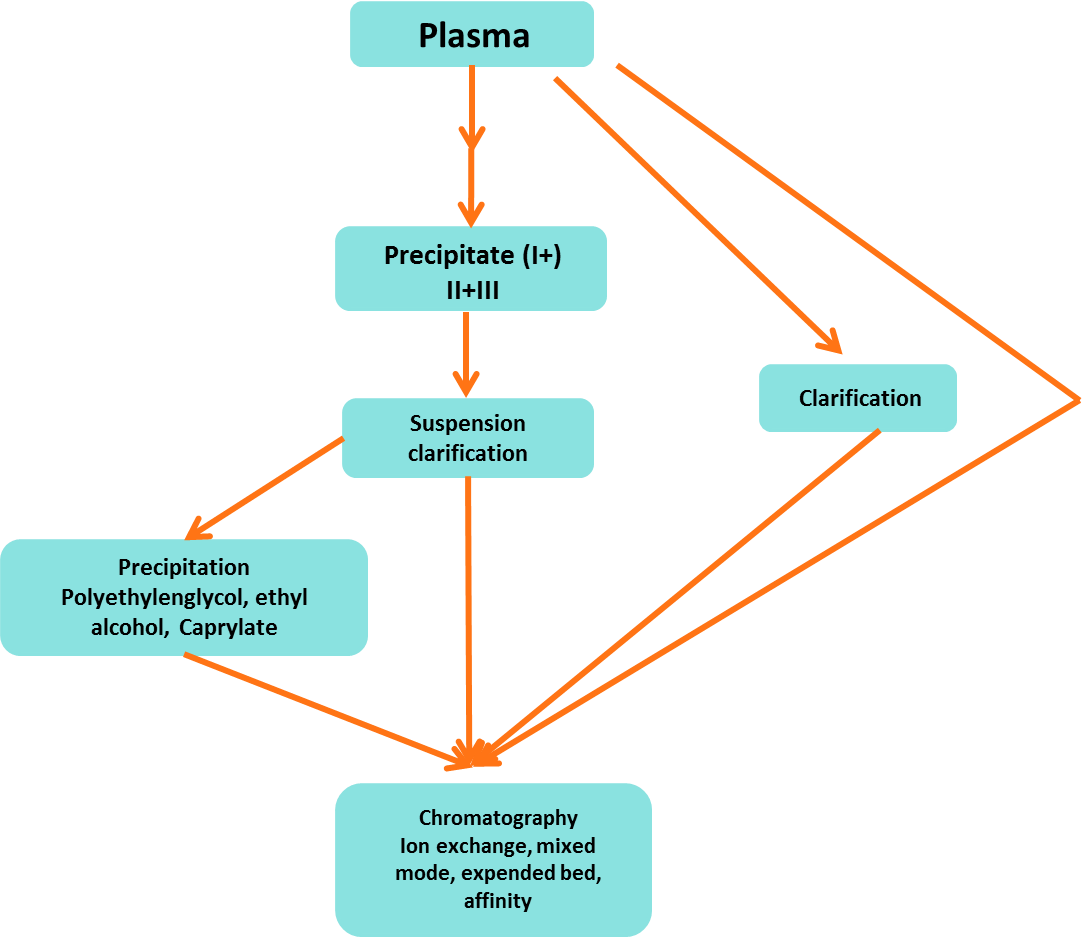
Figure 1: Old fractionation method for IgG separation from human plasma
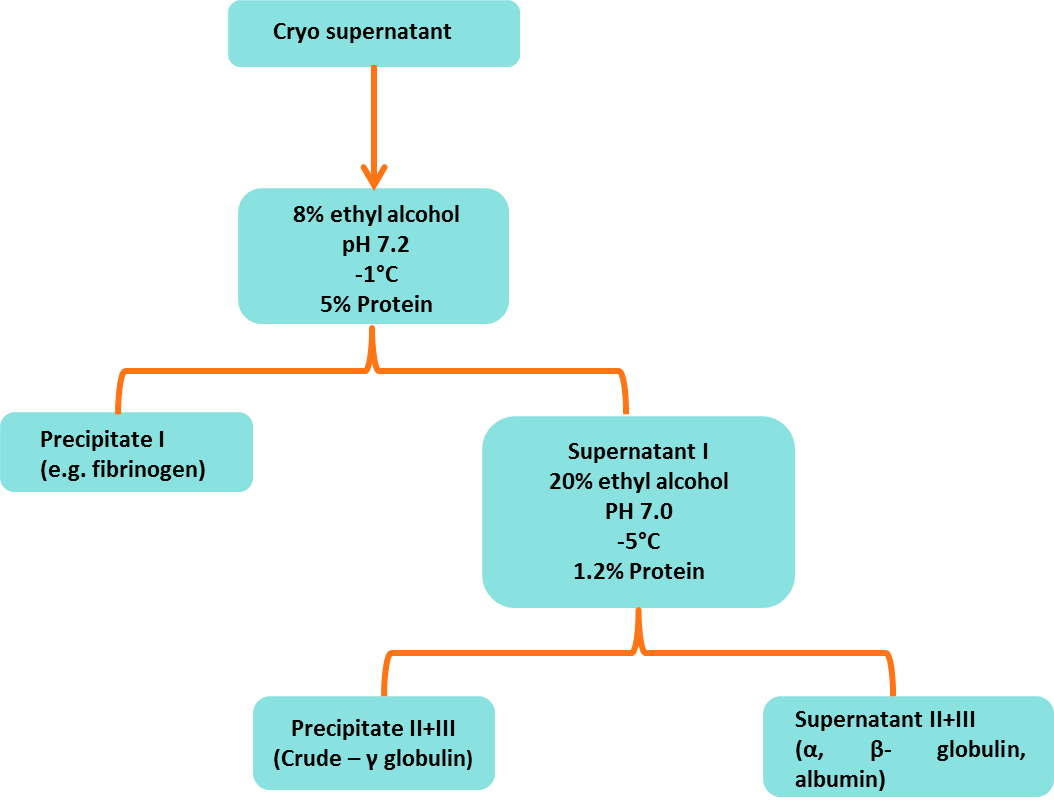
Figure 2: Modern separation Technique for human IgG
4. Overview of Conventional Methods
4.1. Protein A Affinity Chromatography
Affinity chromatography removal can be achieved by a many number of process. The most popular approaches are combinatorial; for example, utilizing Protein-A Affinity Chromatography followed by anion-exchange chromatography. Affinity chromatography have been the predominate method of antibody purification because of its high selectivity. Many different forms of affinity chromatography using bacterially derived receptors such as staphylococcal Protein-A (SpA), streptococcal Protein-G, Protein-L from Peptostreptococcus magnus and Protein-M from Mycoplasma. Protein-A chromatography is the method of capture and purification of therapeutic IgG and Fc-fusion proteins on an industrial scale. Protein A Affinity Chromatography binds to the Fc region of IgG with high specificity. They have some limitations such as high cost of resin, not suitable for all IgG subclasses or species and harsh elution (low pH) may denature IgG.
4.2. Cold Ethanol Fractionation (Cohn Process)
Cold ethanol method also known as cohn process. This method is developed by Edwin J. Cohn in the 1940s. They carried out plasma fractionation at low temperature by addition of ethanol from 8% to 40% V/V at the end for separation of albumin. In Cohn's method have five fractions of obtained which are from fraction I to fraction V. fraction I to V was prepared by adjusting parameters such as the concentration of ethanol, concentration of protein, temperature and pH. In the concentration of ethanol from 8% reaches to 40% in preparation of fraction V there have some limitations like co-precipitation of other proteins (e.g., albumin, IgA, IgM) requires low temperature (cold rooms) and High ethanol consumption which is toxic and flammable. In this method the high purity and yield of Human IgG is not clearly obtained.
| No. of Fraction | % Protein | Components |
|---|---|---|
| I | 5-10 | Fibrinogen, Clotting factor VIII |
| II+III | 25 | IgG, IgA, IgM |
| IV-1 | 5-10 | α- and β-globulins, α₁-antitrypsin, Antithrombin III |
| IV-4 | 5-10 | Complement components, Ceruloplasmin, Haptoglobin, Transferin |
| V | 50-60 | Albumin |
Table 1: Percentage of the main proteins in different fractions archived by plasma fractionation
4.3. Ion Exchange Chromatography
Ion exchange chromatography is also known as Anion/Cation-exchange chromatography. In this method basically relies on the Pi Value of the protein to achieve separation. In the process of cation-exchange chromatography, a positively charged protein is attracted to a negatively charged solid support. In anion-exchange chromatography a negatively charged protein is attracted to a positively charged support. In Ion-exchange chromatography can be used for fully substitute costly Protein-A resin for mAb purification. This is very useful technique for purify for Good polishing step after initial purification and can remove DNA, host cell proteins and some aggregates. They have some drawbacks such as not used for IgG specific antibody, requires optimization of pH and salt concentration and Not ideal for direct capture from complex mixtures.
4.4. Ammonium Sulfate Precipitation
Precipitation technique is using for ammonium sulphate. It is the one of the oldest method in which they are widely applicable approach for partial purification of antibodies. In this solution, a large number of water molecules are bound to the sulphate ion for the reducing the amount of water available to interact with the protein molecules. At a particular concentration (NH₄)₂SO₄, in this insufficient quantity of unbound of water will remain to keep a given protein species in solution and resulting in precipitation. A saturated solution of ammonium sulphate is added to serum or ascites to precipitate the antibody. Ammonium sulphate is useful for polyclonal and monoclonal IgG isolation and a solution with 35% saturated will produce a pure IgG precipitation, but not all IgG will be contaminated with other proteins (eg., albumin). Precipitation at 45% saturated produces an ideal starting material for other purification techniques. This method helps to concentrate IgG and separate it from other serum proteins based on differential solubility. Only 60-80% yield is pure of IgG is obtained by producing on large scale of industrial process recovery of IgG.
4.5. Gel Filtration (Size Exclusion Chromatography)
Gel filtration chromatography is also known as size-exclusion chromatography (SEC). This purification of IgG has been widely used. This technique used for purifying and analyzing human IgG (Immunoglobulin G) based on molecular size. SEC with dextran, agarose and polyacrylamide soft gels was one of the first techniques developed for the purification of proteins. However, Soft gels cannot tolerate high pressure and required long separation times. In SEC technique separates molecules based on size (hydrodynamic volume) as they pass through a column packed with porous beads. They also have limited stability to extremes of pH and to salt. And other limitations of "conventional" size-exclusion chromatography are responsible for the problems with resolution often observed when analyzing/purifying IgG using such methods.
5. Novel Techniques for Purification of Human IgG
5.1. Membrane Chromatography
Membrane chromatography effectively combines the liquid chromatography of high resolution with the membrane of high throughput. Membrane chromatography purifies novel human IgG by using a functionalized porous membrane to selectively bind the IgG, allowing for high-speed capture and purification. Membrane chromatography has high flow rate and lower the pressure drops and in disposable formats. This method is used in Effective in flow-through mode for polishing steps to remove IgA, IgM and aggregates. This process is used high recovery of IgG in industrial scale and purified 98-99%.
5.2. Aqueous Two-Phase Systems (ATPS)
Aqueous two-phase system (ATPS) is a liquid-liquid fractionation technique and has gained an interest because of great potential for the extraction, separation, purification and enrichment of proteins, membranes, viruses, enzymes, nucleic acids and other biomolecules both in industry and academia. The ATPS is able to give high recovery yield and is easily to scale up. It is also very economic and environment friendly method. The use of affinity ligands in ATPS can result in the higher recovery yields and higher purification folds of biological products as it is a primary stage recovery technique. The protein recovery from crude feedstock's at large-scale has been done by ATPS and this application of ATPS has attracted the most interest. ATPS is a simple, selective and low cost promising separation technique. Easy scalability of this technique makes it valid to be adopted by industries for downstream processing.
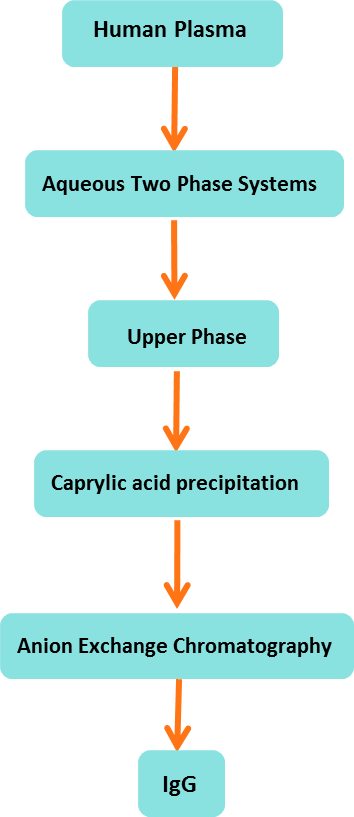
Figure 3: Flowchart of extraction of IgG by aqueous two phase system
5.2.1 Factors Influencing Partitioning in ATPS
Molecular weight (MW) and concentration of polymer: As most of the ATPSs are composed of polymer-polymer/salt. MW of polymers greatly influences the partition. Generally:
- ↑ MW of polymer → ↓ concentration of polymer required for phase formation
- ↑ Differences between the MW of polymers → ↑ asymmetrical curve of the phase diagram
- ↑ MW of PEG → ↓ value of K
5.3. Monolith-Based Chromatography
Monolith-based chromatography were first introduced by Nobel Prize winner Richard Synge, in 1952. This method is used for separating protein from particulates using a "continuous block of porous gel". In chromatographic terms and condition a monolith is defined as a porous, single unit material incorporated into a chromatographic device. The manufactures and analytical laboratory have been interested in the use of Monolith-Based Chromatography because of associated low cost, high porosity and ease of use.
5.4. Mixed-Mode Chromatography
Mixed-mode chromatography has been focused as a cost-effective new technique for antibody purification. Immunoglobulin G (IgG), bovine serum albumin (BSA) and the binary mixture of BSA and IgG were used as the model feedstock to compare the separation behaviors by pH gradient elution. The comparison analysis showed mixed-mode resin with N-benzyl-N-methyl ethanol amine as the ligand had the best ability to separate IgG and BSA. The results indicated that mixed-mode chromatography would be a potential option for antibody purification with the control of loading and elution conditions. A mixed-mode ligand, 4-(1H-imidazol-1-yl) aniline, was coupled to Sepharose Fast Flow to fabricate AN SepFF adsorbents with ligand densities of 15-64 mmol/L, and the chromatographic performances of these adsorbents were thoroughly investigated to identify a feasible approach to improve immunoglobulin G purification.
5.5. Hydrophobic Interaction Chromatography (HIC)
HIC separates antibodies based on their hydrophobicity, facilitating purification under a mild condition without any denaturation. This method is more particularly useful for separating antibodies from contaminates with similar charges.
6. Comparison and Selection of Purification Technologies
| Parameter | Affinity Chromatography | Ion-exchange Chromatography | Hydrophobic Interaction Chromatography | Reverse Phased Chromatography | Gel Filtration Chromatography |
|---|---|---|---|---|---|
| Mechanism | Specific binding | Charge interactions | Hydrophobic group interactions | Molecular size | Hydrophobicity differences |
| Selectivity | |||||
| Capacity | |||||
| Speed | |||||
| Resolution | |||||
| Recovery Rate | |||||
| Initial Conditions | Specific binding conditions; Sample volume unrestricted | Low ionic strength; Sample volume unrestricted | High ionic strength; Sample volume unrestricted | Restricted sample volume and flow rate | Sample volume unrestricted; Additives may be required |
| Termination Conditions | Specific elution conditions; Concentrated sample | High ionic strength; Concentrated sample | Low ionic strength; Concentrated sample | Buffer exchange (if needed); Diluted sample | Risk of biological activity loss in organic solvents |
| Disadvantages | Requires specific affinity reagents and ligands, higher cost | Needs condition optimization for different antibodies, more complex operation | High salt loading required, harsh elution conditions | Low separation efficiency, small processing capacity, can't concentrate samples | Prone to protein denaturation, requires specific equipment support |
| Capture | ★★★ | ★★★★ | ★★★ | ★ | - |
| Intermediate Purification | ★★★ | ★★★★ | ★★★★ | ★ | ★ |
| Polishing | ★★ | ★★★★ | ★ | ★★★★ | ★★★★ |
Table 2: Comparison and selection between Purification technologies
7. Key Challenges in Recombinant IgG Expression
The creation of recombinant immunoglobulin G (IgG) is essential for the development of therapeutic antibodies. However, recombinant IgG expression has several difficulties that may affect functionality, yield and purity.
7.1. Chain Assembly and Pairing
Recombinant IgG formation is very complicated process by pairing with heavy and light chains. They are not in paired with chain byproducts and complicating with downstream processing. And Low Levels of impaired of byproducts occur by despite chain pairing methods. Contaminants are closely resembled by the desired product make purification in difficult.
7.2. The Formation of Byproducts
Multiple co-expressing polypeptide chains can cause aggregates and light chain impairing and heavy chains are homodimers. Because they resemble target IgG, these byproducts are hard to remove in formation. They originated from uneven chain expression and improper assemblies. Purification methods must be effective for removing these pollutants from the byproduct of formation and they effect on purity of human IgG purification method.
7.3. Limitations of the Expression System
Expression system selection is crucial to recombinant IgG production. In Chinese hamster ovary (CHO) cells retain some intracellular IgG in the endoplasmic reticulum even when fully assembled. This retention reduces secretion and yield of Expression System and Cellular bottlenecks must be identified and solved to optimize production.
7.4. Glycosylation Difficulties
IgG function depends on proper glycosylation and Achieving a suitable and consistent glycosylation patterns in recombinant systems can be difficult. Changes in glycosylation can impact the antibody's immunogenicity, stability and effectiveness. Solve these problems sophisticated methods are being investigated such as adding particular glycosylation sites.
7.5. Bispecific Antibody Production
Bispecific antibodies require several chains, making them more complicated. Avoiding impairing and correcting heavy-light chain pairing is a major concern. Because mismatched chains might create pollutants that are hard to extract from the desired product, purifying methods are needed to eliminate these byproducts.
7.6. Complexities of the Purification Process
Contaminants and byproducts complicate purification. Some pollutants are hard to remove and impair yield because they closely imitate target IgG. Researchers are developing novel IgG cleaning methods to improve stability and efficacy. Mixed-mode chromatography and cleaning components are one way.
8. Limitations of Traditional Purification Methods
While conventional approaches have been the industry standard for decades, they are not without drawbacks:
- Cohn Cold Ethanol Fractionation: Uses of many organic solvents and requires low temperatures making it energy intensive and less environmental friendly.
- Ion Exchange Chromatography: Ion exchange Chromatography have limited lack of specify and often requiring multiple steps for sufficient purification.
- Protein A Affinity Chromatography: Protein A Affinity Chromatography having Highly selective for IgG but this process is costly and protein A leaching poses product contamination risks.
- Polishing Steps: Often fail to consistently remove trace levels of closely related immunoglobulin contaminants.
9. Future Outlook
The future of human IgG purification will be shaped by continuous improvements in established technologies and methods for driven by the increasing demand for antibody therapies and the need for more efficient and cost-effective processes. There is increasing regulatory pressures and demand for higher purity biologics. A Novel purification platform will become the industry standard. And integration with industry practices including automation, a real-time analytics and digital process control and it will further have optimized by downstream processes. In advances a synthetic biology and machine learning will aid in the design of custom ligands and dynamic process optimization strategies. And in future focuses on the growth of the market and new methods or techniques for purification of human IgG in which they are cost effective and produce high purity rate and yield of IgG with less amount of steps can done for one process.
10. Conclusion
The biopharmaceutical industry stands at a critical junction in whom traditional IgG Purification methods are no longer sufficient to meet growing demands and regulatory expectations. In Novel purification techniques provide a pathway toward more efficient, scalable and sustainable processes. These innovations are essential and important not only for ensuring product quality and compliance but also enabling broader access to life-saving IgG therapies globally. In this we compare the novel methods of purification of human IgG how they are done with in few steps with less amount of time and producing high yield and purity of Human IgG antibody at a large industrial scale because in old traditional method is good for laboratory but in large scale they contained so many contamination and not producing high purity of human IgG. Further we can see that traditional methods have some limitations in which they use so many solvents, done multiple steps for one process and they are very costly at a large scale also in the end they not produce high yield of purification. In novel methods focusing on cost, less multiple steps and use less amount of solvents and they producing high purity of IgG. It is investing in modern purification technologies is not just an upgrade but it's a strategic imperative for the future of antibody therapeutic.



-Sensors-Next-Generation-Flexible-Devices/Wrinkled-Silver-Nanowire-AgNW-Sensors-Next-Generation-Flexible-Devices.png)


