1. Introduction
One of the most recent advancement in the medical biology is Messenger RNA (mRNA). The highly successful vaccine for Covid-19 pandemic brought mRNA to a global acclaim. Recognizing this brilliant advancement, the Nobel Prize in Medicine 2023 was bestowed to Katalin Karikó and Drew Weissman for their work in modifying mRNA leading to its clinical use. Traditional therapies typically focus on blocking or supplementing proteins through chemical compounds or biologics. However, mRNA leverages the body’s own cellular machinery to provide with therapeutic proteins, enabling the body to combat various diseases effectively. This distinctive mechanism has prompted scientists to reconsider their approach to treating various diseases, establishing a basis for advancements in genetic disorders such as allowing cells to correct proteins associated conditions like cystic fibrosis or hemophilia, regenerative medicine, and cancer therapy. [1] These advancements signal a shift from managing symptoms to addressing underlying causes, improving treatment outcomes and expanding healthcare possibilities.
The foundational idea of mRNA therapeutics is quite simple. Delivery of mRNA sequence to cell, further leads to synthesis of proteins encoded as if it were naturally part of it. Theoretically, any protein can be synthesized with the use of this method. Yet, in reality mRNA comes with two big hurdles. First is its high immunogenicity and second being the presence of RNAse almost everywhere. The nucleotides in the mRNA were modified putting an end to these issues leading to the development of Covid-19 vaccine.
| Aspect | Traditional Therapeutics | mRNA Therapeutics |
|---|---|---|
| Mechanism of Action | Directly delivers proteins, small molecules, or antibodies | Uses synthetic mRNA to instruct cells to produce proteins |
| Target Versatility | Limited to specific protein interactions or pathways | Can theoretically target any gene or protein |
| Safety | Some therapies (e.g., gene therapy) may integrate into DNA | Does not integrate into genome, reducing mutagenesis risk |
| Manufacturing | Complex production—drugs require cell cultures | Easier to scale—mRNA can be synthesized quickly |
| Delivery Method | Often oral, injectable, or intravenous with established methods | Requires lipid nanoparticles (LNPs) for stability |
| Applications | Cancer treatment, antibiotics, monoclonal antibodies | Cancer immunotherapy, rare genetic disorders, vaccines |
| Storage Requirements | Typically stable at room or refrigerated temperatures | Requires ultra-cold storage for stability |
Despite the promise of mRNA-based treatments, challenges remain, particularly in ensuring stability, refining delivery systems, and streamlining regulatory approvals. Advances in lipid nanoparticle formulations have improved mRNA delivery, but ongoing research continues to optimize efficiency and safety [3]. Regulatory frameworks must also evolve to accommodate the rapid development of mRNA therapies, ensuring timely access for patients in need.
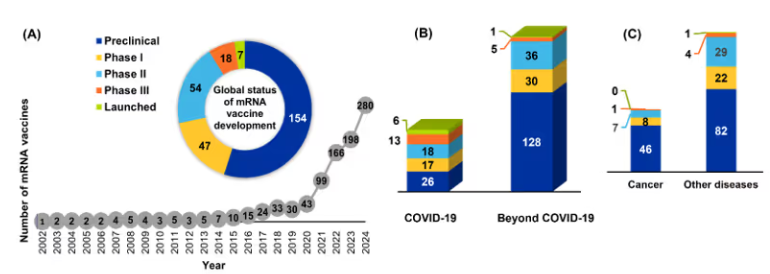
Figure: (A) Line graph shows active mRNA vaccine preclinical and clinical trials over the years and the pie chart shows the global status of mRNA vaccine development. (B) Number of preclinical and clinical trials of mRNA vaccines on COVID-19 and beyond COVID-19. (C) Distribution of mRNA vaccine trials among cancer and other diseases.
Compared to traditional therapeutics, such as small-molecule drugs and protein-based biologics, mRNA offers rapid adaptability and personalized treatment options. Conventional drugs often target enzymes or receptors through complex biochemical interactions, while biologics require complicated manufacturing processes to produce specific proteins externally. mRNA therapies, however, bypass these limitations by enabling the body to generate therapeutic proteins directly—leading to faster development and more precise measures. This difference makes mRNA a game-changer in modern medicine, setting the stage for transformative advancements across multiple disciplines.
2. Innovations in mRNA Therapeutics
Messenger RNA (mRNA) therapeutics is getting a lot of attention as it has the ability to improve the way therapies are conducted. Its ability to enter a cell and then produce protein as its own has gathered a lot of attention. This technology has changed the way therapies were being performed making them more patient specific causing negligible or very low side effects as well as increasing the chances of acceptance of proteins by the body.
2.1 Protein Replacement Therapy
Protein replacement therapy using mRNA holds great promise for treating genetic disorders but faces challenges like high dosage requirements, immunogenicity, and targeted delivery. Advances in nucleotide modifications and delivery methods are crucial for its broader clinical application.
Protein replacement therapy is a medical approach designed to restore essential proteins that are missing or malfunctioning due to genetic mutations. By supplying these proteins, either directly or through advanced methods like mRNA-based therapies, this treatment aims to help patients regain normal biological functions and improve their quality of life. It holds promise for addressing a range of genetic disorders, offering new hope where traditional treatments may be limited [7]. One of the biggest challenges faced by therapeutic application of mRNA is need for much higher dosage which may sometimes require lifelong treatment. This necessitates large-scale mRNA production and stable, long-term expression in vivo [8]. While some immune activation is beneficial for vaccines, it can certainly be problematic in protein-replacement therapies that can be mitigated through nucleotide modifications and stringent purification processes. Additionally, effective delivery remains a challenge, as mRNA tends to accumulate in the liver when formulated with lipid nanoparticles (LNPs). Many diseases require tissue-specific expression, making targeted delivery crucial—especially for therapies like CAR-T treatments or cytokine-based cancer therapies. Overall, while mRNA technology holds great promise for protein-replacement therapy, overcoming these challenges will be essential for broader clinical applications [8].
2.2 Passive Immunotherapy
mRNA technology is modifying passive immunotherapy continuously by empowering the body to produce antibodies, cytokines, and immune checkpoint regulators on its own. By bypassing the complexities of traditional protein expression in a lab, this approach speeds up treatment development while making it more accessible and cost-effective [8].

2.3 Enhancing protein yield
One of the advancements in mRNA therapeutics is the use of chemically modified nucleosides, particularly in uridine moieties, to enhance protein expression in cells. These modifications play a crucial role in reducing immune system detection, allowing for significantly higher efficiency in mRNA translation. The introduction of methylpseudouridine has been especially transformative, enabling mRNA to evade immune sensors like Toll-like receptors, boosting protein expression up to 100-fold compared to unmodified strands. With over 130 naturally occurring RNA modifications, researchers are continuously exploring new combinations to refine delivery methods, stability, and effectiveness. Shifting from complete nucleoside substitution to partial modifications, a pattern seen in naturally synthesized mammalian mRNAs presents exciting possibilities for further optimization [9].
2.4 Gene Therapy
mRNA-based gene editors have a great impact on medicine by enabling precise genetic modifications for treating disorders like muscular dystrophy and glioblastoma.
mRNA has emerged as a promising tool for transporting programmable nucleases, enabling precise gene regulation in living organisms and laboratory environments. This approach holds immense promise for advancing gene-editing treatments. Traditional gene editing methods focus on creating targeted insertions or deletions and mutations by inducing a double-stranded break in DNA. In contrast, mRNA-based gene editing therapeutics take a different approach, rather than directly modifying the DNA sequence, they enable precise programming and cellular function repair by encoding artificial nucleic acid endonucleases. CRISPR/Cas9 has gained the most attention due to its simplicity, efficiency, and ease of design, making it one of the most accessible tools in modern gene editing [10].
Recent advanced modifications in mRNA-based gene editing therapies are opening new doors for treating genetic disorders and certain cancers. For muscular dystrophies—monogenic diseases that lead to progressive muscle degeneration—scientists have achieved up to 90% genome editing efficiency in human primary muscle stem cells from multiple donors using mRNA-delivered SpCas9 and adenine base editors. Similarly, synthetic mRNA-based gene therapy is emerging as a promising treatment for glioblastoma (GBM). Studies have successfully tested TRAIL-mRNA and PTEN-mRNA in GBM cells and xenograft animal models, demonstrating the potential of this therapeutic strategy. With the flexibility and ease of handling synthetic mRNAs, this approach is paving its way for personalized treatments for GBM patients, pushing the boundaries of gene editing and regenerative medicine [10].
3. Expanding Horizons: mRNA in Medicine
Messenger RNA (mRNA) technology has advanced far beyond its role in COVID-19 vaccines, opening many doors in medicine. With its ability to instruct cells to produce specific proteins, researchers are now more focused in cancer treatment, genetic disorders, genetic disorders, autoimmune conditions, and personalized medicine. This technology is modifying the way diseases are treated, offering potential solutions that traditional methods could not achieve as efficiently.
3.1 Cancerous diseases
mRNA-based cancer immunotherapy is transforming treatment by enabling targeted strategies like therapeutic vaccination, adoptive cell therapy, humoral immunotherapy, and immuno-epigenetic modulation. These approaches show promise in clinical trials for various cancers, offering new hope for patients.
In 2020, around 19.3 million cases were diagnosed of cancer, and approximately 10 million people have died of cancer worldwide according to Global Cancer Statistics [4]. One of the major developments in mRNA medicines is its role in cancer immunotherapy. Due to mRNA technology’s flexible nature, it can support wide range of anti-cancer treatments extending from tumoral antigens to monoclonal antibodies. mRNA-based therapeutic strategies that have been clinically used for cancer can be categorized in four categories; Therapeutic vaccination, Adoptive cell therapy, Humoral immunotherapy, and Immuno-epigenetic modulation. These strategies are now being clinically tested and showing promising results [5].
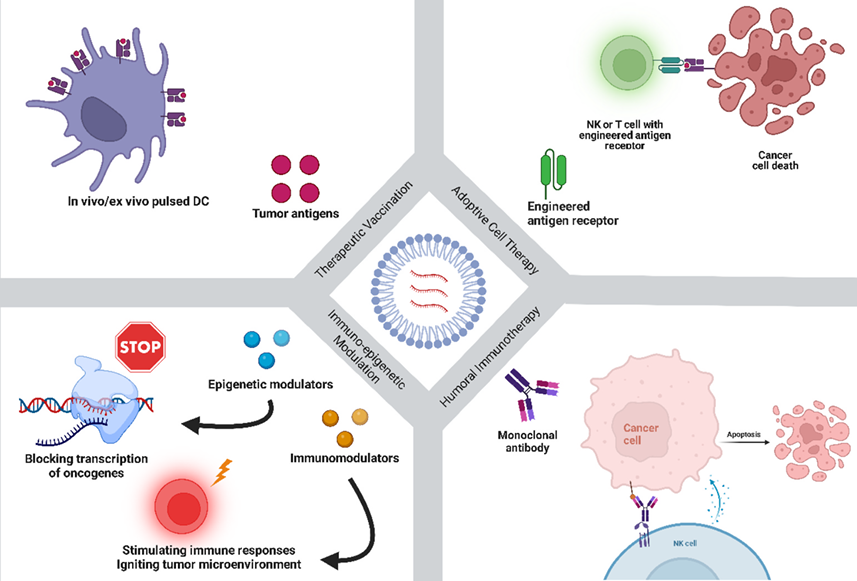
- Therapeutic Vaccination: It is the most widely used mRNA based strategy for tumors, that deals with in-vivo and ex-vivo loading of Antigen presenting cells (APCs), specifically dendritic cells (DCs) with whole mRNA, synthetic mRNA encoding tumor encoding antigens. These pulsed DCs further interact with T cells leading to eradication of tumors.
- Adoptive cell therapy: It may be explained as the delivery of natural or modified immune cells, such as Natural Killer (NK) cells, tumor-infiltrating lymphocytes (TILs), T cells, and macrophages inside the body of patients to eliminate cancer cells as an immunotherapeutic approach [5].
- Humoral immunotherapy: Many FDA-approved monoclonal antibodies are now being used for the cancer immunotherapy, they only enjoy partial acceptance. mRNA technology can be proved as an alternative that has the ability to overcome most these challenges. It can be solved by the delivery of formulated mRNA into the patient’s body. This mRNA can yield high amount of proteins and at the same time minimize the injection-dosing frequencies, sudden exposures, and their related side effects.
- Immuno-epigenetic modulation: It is a strategy where mRNA delivers epigenetic controllers, immune and cytokines to suppress the oncogenes, activating the immune system, and further reprograming the tumor environment. Recently, mRNA-2752 has been used for treatment of solid tumors as monotherapy or along with Durvalumab was effective in suppression of tumor.
All these strategies are now being tested in clinical studies for various types of cancer like pulmonary osteosarcoma, CLDN18.2- positive solid tumors, lymphoma, liver cancer, Leukemia, etc. [5].
3.2 Auto-immune diseases
Autoimmune diseases are a rare kind of diseases where the body’s immune system mistakes its own body tissues as foreign bodies and give an immunogenic response for it. mRNA therapies can play a major role to re-educate these immune cells by many different ways. Some of these include regulation of overactive immune pathways, tolerance to self-antigens, and supplying deficient proteins which are now under investigation. Early research on multiple sclerosis and type 1 diabetes has shown encouraging results hinting a possible modification on how immune system works [6].
3.3 Infectious diseases
The COVID-19 pandemic highlighted the potential of mRNA technology in rapidly responding to infections. One of its biggest strengths is the ability to be quickly modified to tackle emerging variants, making it invaluable in high-mutation pandemics. Beyond its speed and cost-effectiveness, mRNA technology has inherent properties that enhance immune response by effectively delivering antigens to T and B cells. While short-term side effects have been minimal, the long-term effects of mRNA-based therapies remain under study. Unlocking these insights will be crucial for expanding its applications across medicine [6].
3.4 Rare genetic disorders
mRNA-based therapies have the potential to transform treatment for rare genetic disorders, many of which stem from mutations that disrupt essential protein production. By delivering precise genetic instructions, these treatments could restore normal function temporarily, offering hope to patients with limited options. Scientists are also investigating ways to use mRNA to correct mutations and regulate gene expression, paving the way for advances in personalized medicine [6].
4. Conclusion and Future Prospects
The success of mRNA-based COVID-19 vaccines has highlighted this technology in medicine, shifting the focus from symptom management to direct cellular intervention. Unlike traditional drugs, which require external synthesis of proteins or small molecules, mRNA unlocks the body's ability to generate its own therapeutic proteins, offering very unique adaptability.
However, despite its promises, mRNA therapeutics face challenges in stability, delivery, immune response, and long-term efficacy. Advances in AI-based mRNA, and self-amplifying RNA are helping overcome these hurdles, paving the way for broader clinical applications.
Looking ahead, mRNA therapeutics will continue to evolve, providing customized treatments, improved stability and efficiency, and an expanded range of applications. The future may hold more advanced modifications and/or treatments being made using mRNA in regenerative medicine, neurological disease treatment, and precision oncology, alongside evolving regulatory frameworks ensuring widespread accessibility.
Challenges & Future Innovations
The mRNA therapeutics is evolving continuously in technology and expanding disease applications. Many techniques and therapies have evolved to minimize the challenges faced by mRNA’s use in therapeutics. mRNA is continuously being modified making it more stable, and easy to deliver. One such example is self-amplifying RNA (saRNA). The evolution of saRNA vaccines offers many advantages over conventional mRNA vaccines by achieving robust immune responses with lower doses. With recent approvals in India and Japan, saRNA-based vaccines are gaining attention, yet further refinements will be crucial for broader adoption. Enhancing their formulation to minimize innate immune overactivation and prioritizing optimized antigen selection—favoring smaller, efficiently expressed sequences—could significantly improve efficacy [11].
Alongside saRNA advancements, AI-powered platforms such as mRNAdesigner are redefining the mRNA sequence optimization. It is an advanced tool that optimizes mRNA sequences by refining coding regions (CDS) and untranslated regions (UTRs) to enhance stability and translation efficiency. Its intuitive interface allows researchers to customize parameters, ensuring precise control over mRNA design for improved protein expression offering a user-friendly platform to refine stability and translation efficiency for therapeutic applications [12]. Future improvements will focus on optimizing modified nucleotides, expanding beyond eukaryotic models, and enhancing mRNA degradation control, paving the way for next-generation therapies.
As research accelerates, mRNA technology will keep evolving, redefining the possibilities for personalized and preventive healthcare in the decades to come.