Executive Summary
Antibody-drug conjugates (ADCs) are used for cancer therapies, improving drug-to-antibody ratios, linker stability, and payload potency. But single‑payload ADCs still face challenges such as tumor heterogeneity, drug resistance, and off‑target toxicity. However, dual‑payload ADCs are emerging as a promising next step by combining two cytotoxic agents within one antibody. This approach helps in broadening therapeutic coverage, reduce resistance, and expand the therapeutic window. While development still remains complex, dual‑payload ADCs represents a major step toward more effective cancer therapies.
1. Introduction
Antibody-drug conjugates (ADCs) are widely used for cancer therapies due to their potential in the delivery of cytotoxins to targeted cancer cells. The first generation of ADC, represented by gemtuzumab ozogamicin, employed antibodies conjugated randomly to cytotoxic agents such as calicheamicin. However, these findings suffered due to unstable linkers, inconsistent drug to antibody ratios (DARs), and restricted control over payload delivery, which eventually led to off-target toxicity. The second-generation ADCs used antibodies, cleavable linkers, and modified conjugation sites which improved DAR consistency and further enabled the use of potent microtubule inhibitors like MMAE and DM1. The third generation ADCs have emphasized on site specific conjugation strategies, use of tumor responsive linkers, and incorporation of next-generation payloads such as topoisomerase I inhibitors e.g., DXd and SN 38.
Despite all these innovations, ADCs still remain burdened by considerable clinical obstacles. Cancer cells develop drug resistance through mechanisms including metabolic irregularities, target antigen loss or mutation, impaired internalization, lysosomal dysfunction, and upregulation of efflux pumps. Recent progress in conjugation methods has given rise to dual‑payload ADCs for the use in combination therapy. Unlike traditional ADCs, which were designed to deliver a single payload to an antigen, dual-payload ADCs are engineered by conjugating two cytotoxic agents onto a single antibody.
2. Problems and Pathways Forward
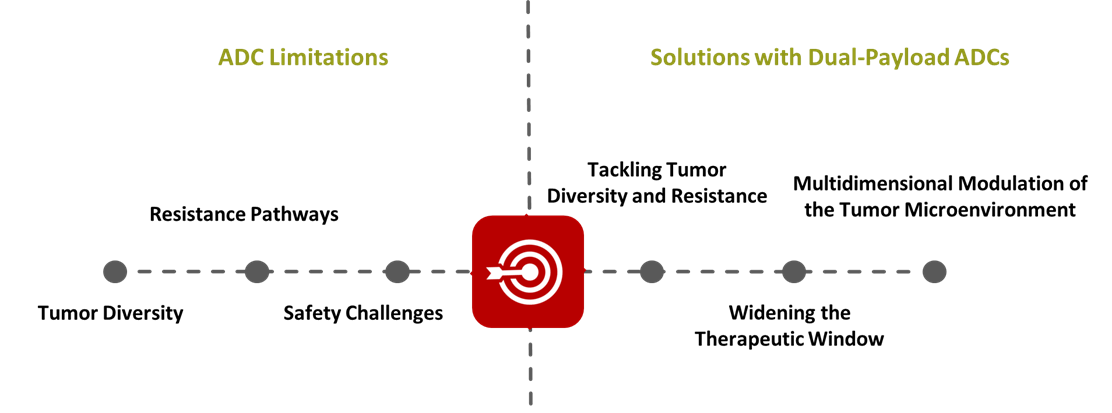
2.1 ADC Limitations: The Roadblocks Ahead
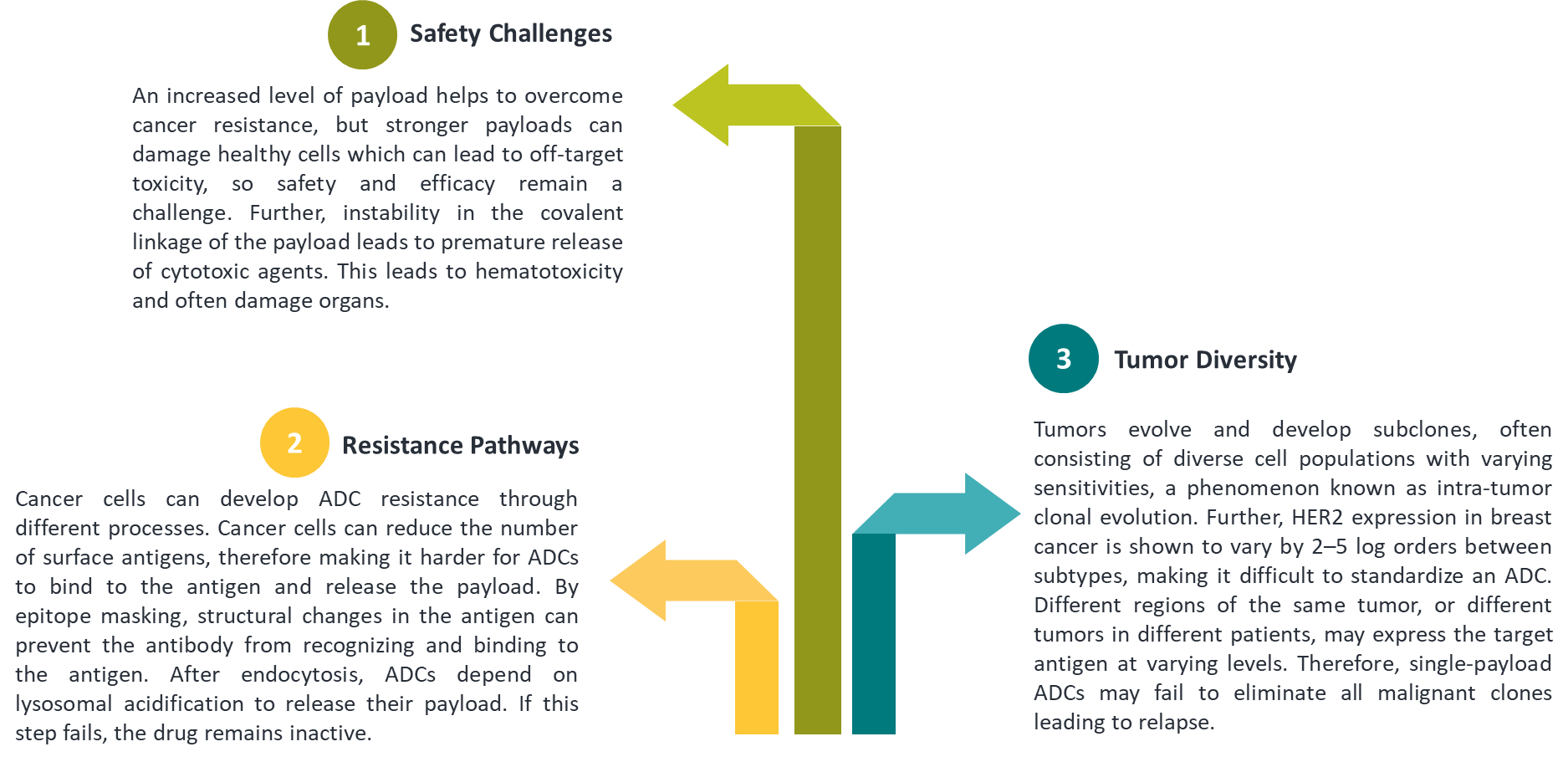
2.2 Therapeutic Solutions with Dual-Payload ADCs

3. Techniques for Dual-Payload ADC Construction
The strategies for generating homogeneous dual payload ADCs can be classified into three main categories, based on the methods of introduction of orthogonality.
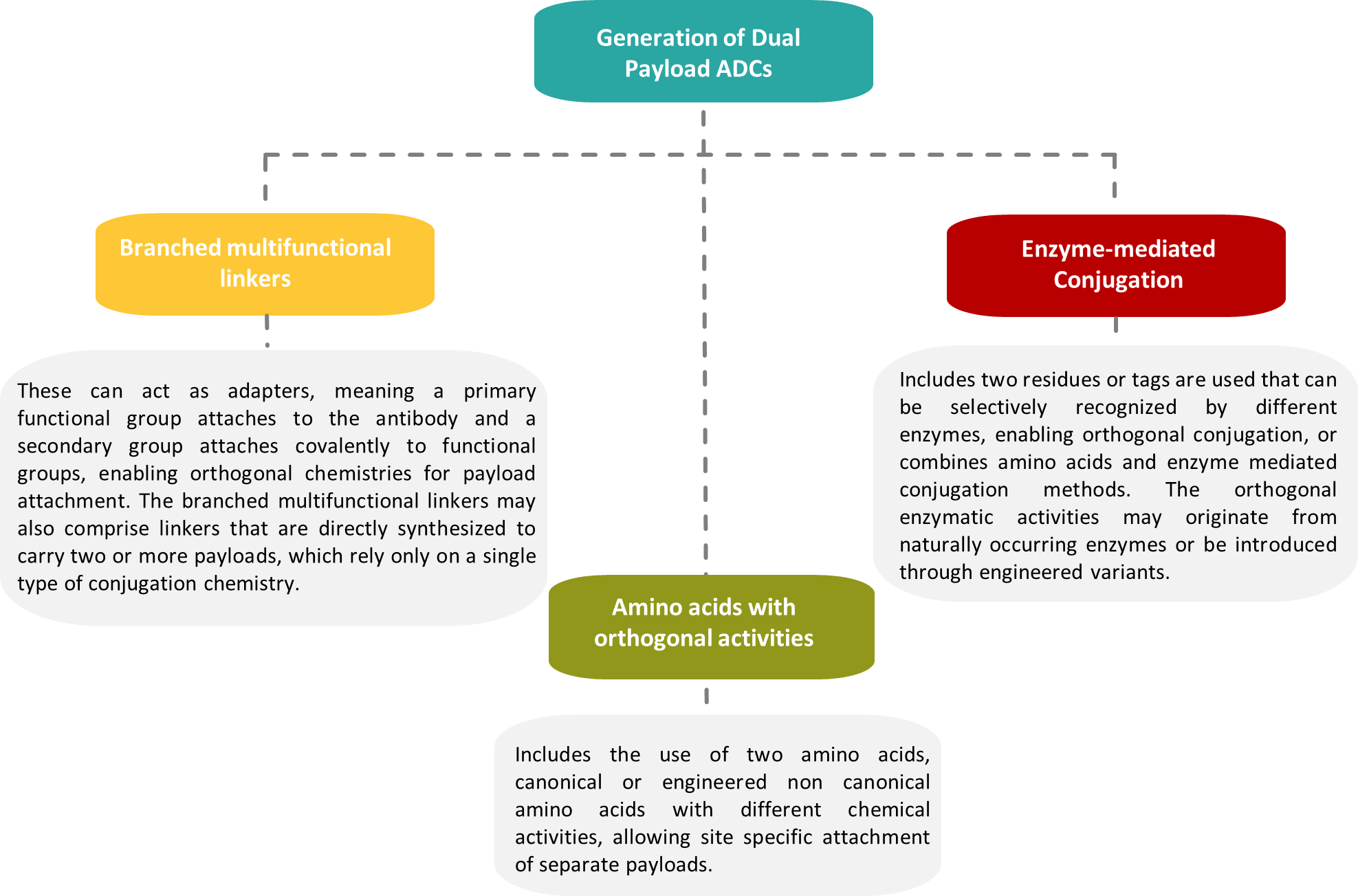
4. Balancing Stability and Release: Linker Technologies

5. IP Activity in Dual-Payload ADCs
This IP analysis focuses on patents related to the synthesis of dual-payload antibody-drug conjugates (ADCs). In total, 199 patent applications were identified, each disclosing methods for constructing dual-payload ADCs.
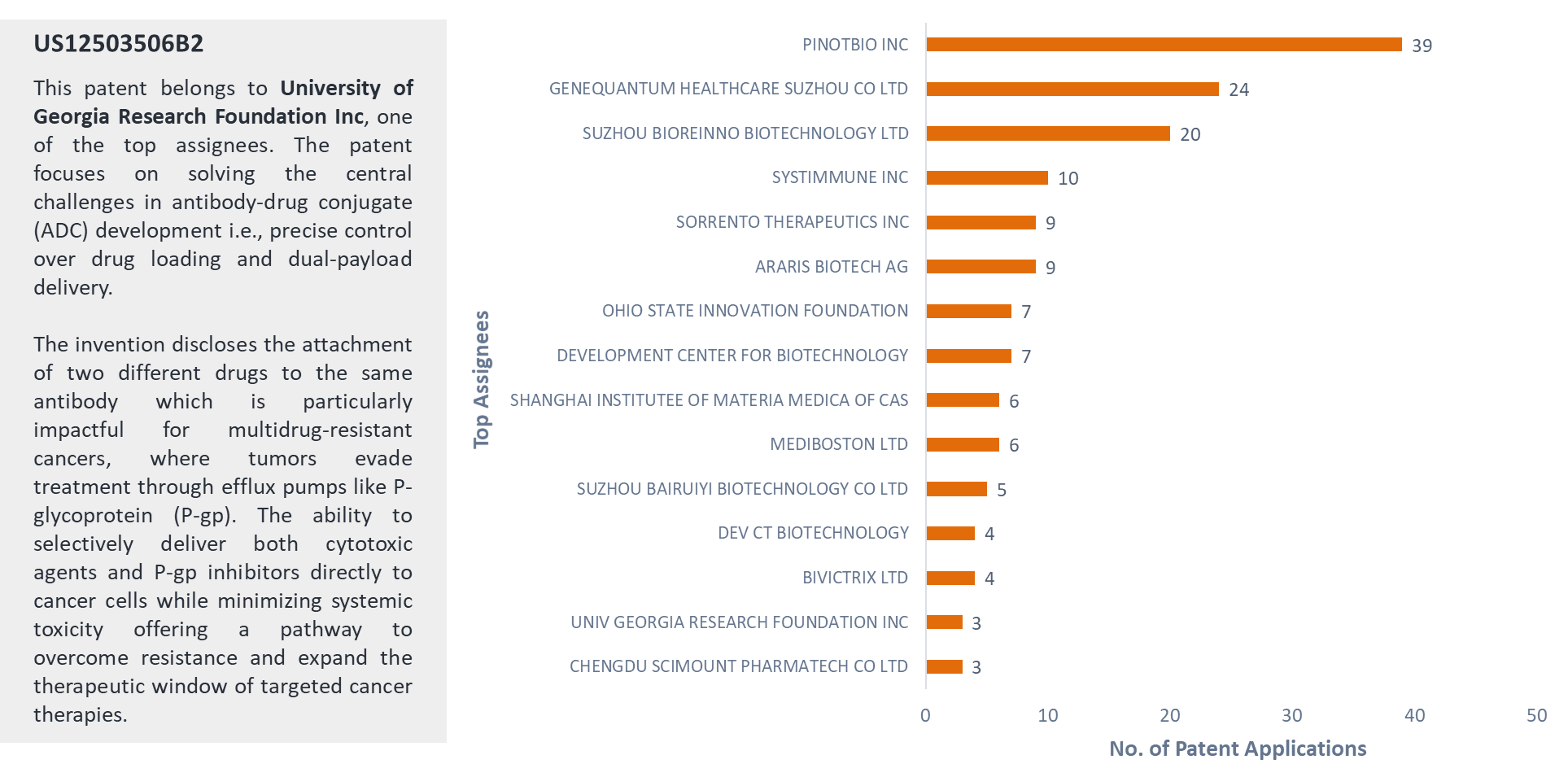
Figure 6 shows the top patent assignees, with Pinotbio Inc leading with 39 patent applications and Genequantum Healthcare Suzhou Co Ltd is second with 24 patent applications.
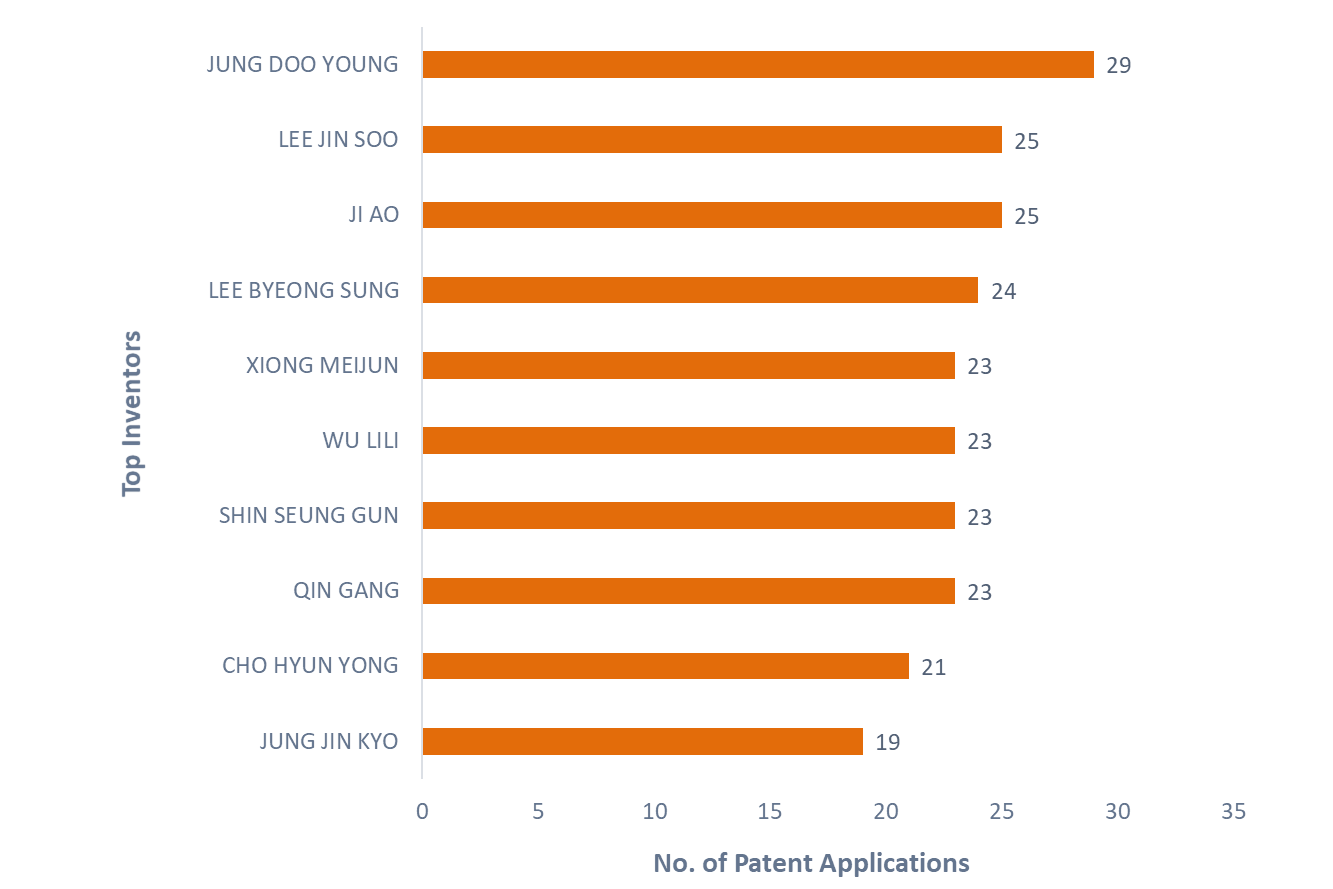
Figure 7 shows the top inventors, with Jung Doo Young leading with 29 patent applications. Lee Jin Soo and Ji Ao are second with 25 patent applications each. Notably, Jung Doo Young and Lee Jin Soo both hold patents assigned to Pinotbio Inc., the top assignee in this patent landscape.
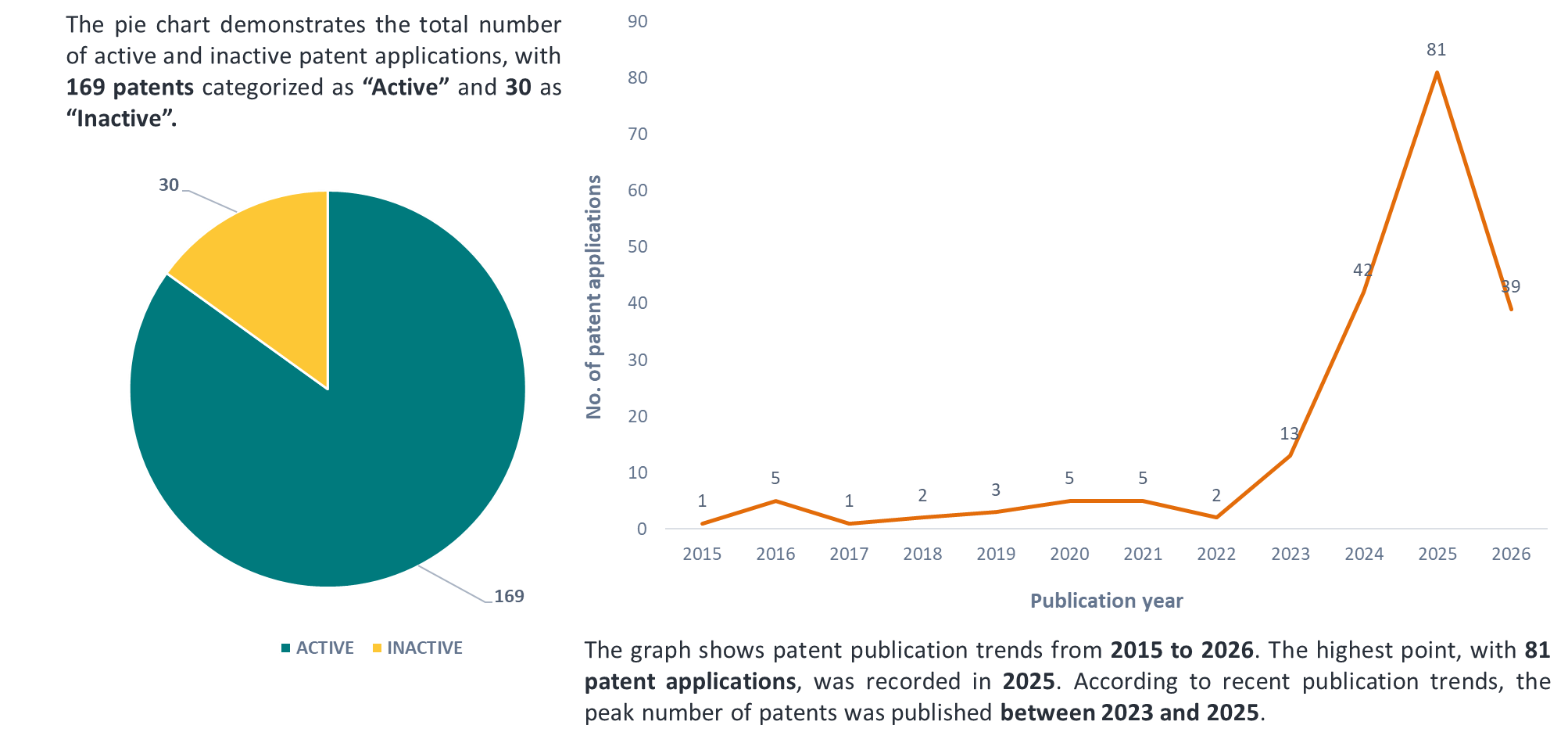
Figure 8 shows a pie chart demonstrating the legal status and a graph demonstrating the publication trends.
6. Market and Commercial Outlook

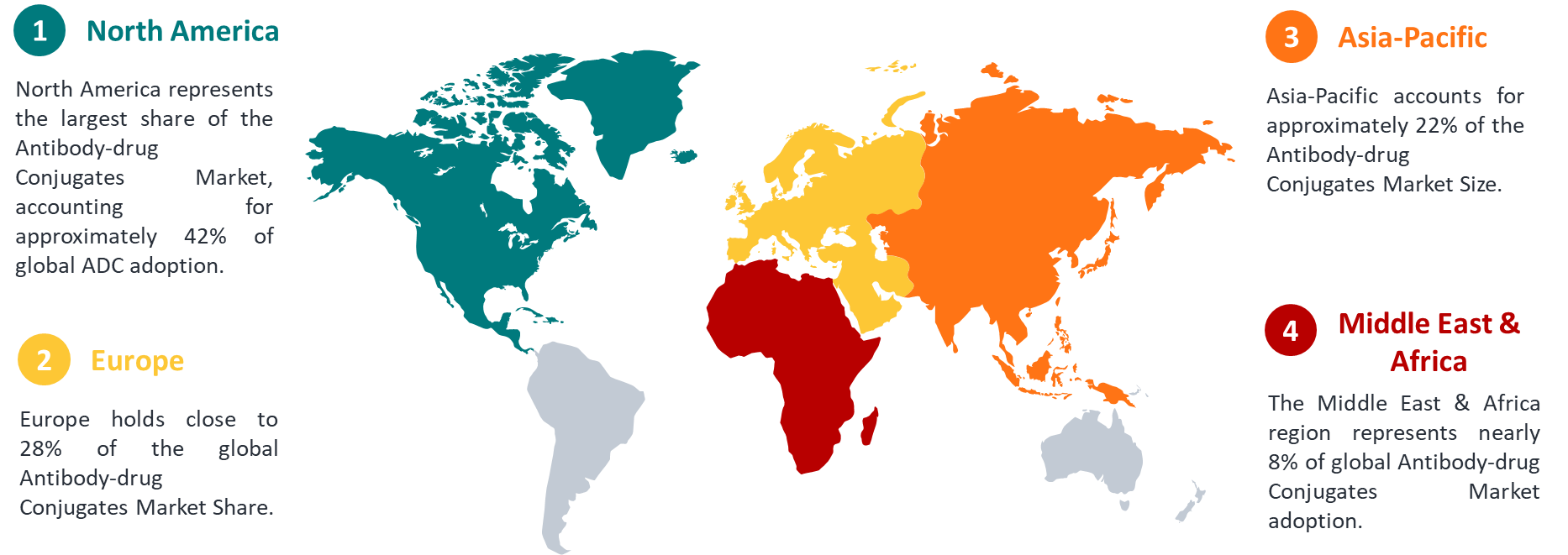
7.1 Antibody-Drug Conjugates Market Regional Outlook
7. Key Challenges in Dual‑Payload ADC Development
Although dual payload ADCs hold considerable therapeutic promise, their development is hindered by numerous challenges.

8. Future Directions in ADC Therapy
Multi payload ADCs are emerging as a promising approach in targeted cancer treatment, with the promise of greater therapeutic efficacy, showing reduced resistance, and improved safety. By combining two cytotoxic agents with distinct mechanisms of action, these constructs show potential to overcome drug resistance, addressing tumor heterogeneity, and delivering more durable responses than single payload ADCs. Advances in technologies such as site specific conjugation, orthogonal linker systems, and non-canonical amino acid incorporation are paving the way for greater precision, stability, and scalability in manufacturing. Dual payload ADCs are expected to open new opportunities beyond oncology, including autoimmune and infectious disease applications, where selective delivery of multiple therapeutic agents could transform treatment procedures. With growing industry investment and early clinical programs already underway, dual payload ADCs are poised to expand into new application areas, offering new scientific innovation with real therapeutic impact.
9. Conclusion
Dual‑payload ADCs provide a multidimensional strategy against cancer, tackling tumor diversity and resistance while minimizing toxicity. Success depends on precise linker chemistry and optimized drug ratios, with innovations like bifunctional and PEGylated linkers already improving performance. Despite challenges in manufacturing, regulation, and safety, dual‑payload ADCs hold strong potential to redefine targeted cancer treatment by combining efficacy with improved control over drug delivery.